
Observational studies help us understand how to treat pandemic influenza and other emerging virus diseases
In his editorial, Zhang (1) comments on my earlier article that suggested inexpensive and widely available generic drugs might be used to reduce mortality from pandemic influenza, Ebola and other emerging virus diseases (2). He notes that several earlier studies explored the possibility that statins might be used to treat patients with sepsis and acute respiratory distress syndrome (ARDS) (1). Many individual observational studies [for example, (3)] and their meta-analyses (4) have suggested that statins might be effective in these patients, but several randomized controlled trials (RCTs) have failed to demonstrate their efficacy (5,6). Critical care specialists, many of them involved in these RCTs, have expressed different opinions on whether statins should still be considered potentially useful (7-9). In my view, the question is still open; the RCTs were conducted in mechanically ventilated ICU patients, and many of these patients were started on treatment late in the course of illness. Thus, statin treatment by itself may have been “too little, too late”. Nonetheless, the experimental and clinical rationale for this general approach to treating the host response in patients with critical illness is still persuasive. For many reasons, it must be further explored (2).
Observational studies with big data are useful
Zhang suggests that observational studies using electronic healthcare records (EHRs), sometimes called big data, could provide insights into potentially useful treatments (1). This is an important idea, although it is not new. Many years ago, I used smaller data sets (then called “linked records” or “administrative datasets”) to explore strategies for hospital-based pneumococcal (10,11) and influenza (12) vaccination and to evaluate the effectiveness of influenza vaccination (13). More recently, much larger administrative data sets have been assembled: for example, the VA Health Care System administrative database in the US that includes clinical data from more than 150 VA hospitals and 850 outpatient clinics (14) and the Clinical Practice Research Datalink (CPRD) in the UK (15). These databases have been used to evaluate the effects of statins and other treatments on outcomes in patients with community-acquired pneumonia (14) and medically attended acute respiratory illness (15). More recently, Chinese investigators have studied the effects of corticosteroid treatment on 30-day mortality among more than 2,000 adolescents and adults hospitalized with influenza A (H1N1) pdm09 virus infection (16). They used clinical data gathered prospectively from 407 hospitals throughout China. Patients who received low- to moderate-dose corticosteroid treatment (25–150 mg/day) had a significant 36% reduction in 30-day mortality, but high-dose treatment had no such effect.
Individually, none of these observational studies should be considered definitive because, like all such studies, they could be compromised by unmeasured confounding. Nonetheless, considered together they strengthen arguments that using these drugs might be beneficial. Moreover, observational studies (more often than RCTs) can suggest new ideas (17). For example, a few years ago it was suggested that statins (which have immunomodulatory effects) might confound estimates of influenza vaccination effectiveness (18). Subsequent studies showed that statins do in fact compromise both the antibody response to (19) and clinical effectiveness of (19,20) influenza vaccination. Paradoxically, in the absence of influenza vaccination, statin treatment appears to reduce the occurrence of laboratory-confirmed influenza (21,22). This latter observation could be critically important when we consider its implications for a global response to the next influenza pandemic, as discussed below.
Observational studies using smaller datasets are also useful
A recent report of a small observational study suggests that corticosteroid treatment, supplemented by a four-day course of intravenous high-dose vitamin C (1.5 g q6h) and thiamine (200 mg q12h) might dramatically improve survival in patients with sepsis (23,24). The study included 47 consecutively treated patients with severe sepsis or septic shock and procalcitonin levels ≥2 ng/mL. Their outcomes were compared with those of a matched patient group treated seven months earlier. In this historical control group, 60% of patients had received hydrocortisone (50 mg q6h) but no vitamin C or thiamine. In the control group, mortality was 40.4%, but in the high-dose vitamin C group it was only 8.5% (propensity-adjusted odds ratio =0.13; 95% CI, 0.04–0.48). The scientific rationale for this study was the known effect of vitamin C in restoring sensitivity to the glucocorticoid receptor, its antioxidant activity and its beneficial effects on endothelial barrier integrity and the microcirculation (23,24). However, its proximate cause was the dramatic improvement seen when this regimen was used earlier to treat three sepsis patients who were on the verge of dying (23).
The recent Ebola outbreak in West Africa has provided another example of a new approach to treatment. Because of clinical similarities between Ebola virus disease and sepsis, in August 2014 it was suggested that statins and other immunomodulatory drugs might be used to treat the host response of Ebola patients (2,25). Ebola scientists, staff of the World Health Organization and other health agencies and foundations involved in the international Ebola response dismissed this idea, and in some instances actively opposed it (2). Nonetheless, thanks to a donation from a private physician, approximately 100 Ebola patients in Sierra Leone were treated with a combination of a statin (atorvastatin) and an angiotensin receptor blocker (ARB) (irbesartan). Some of these patients also received a three-day course of clomiphene, a selective estrogen receptor modifier known to have antiviral effects against Ebola virus. Only three inadequately treated patients are known to have died (2,26,27). Local physicians who treated these patients refused to publicly release information on their experience, but their letters and memoranda provided convincing evidence of patient benefit.
Unfortunately, articles that have reviewed the clinical trials of experimental Ebola treatments have ignored the experience in Sierra Leone (2,28,29). In one instance, it has been dismissed as an anecdotal report of survival from which “it is impossible to draw any meaningful conclusions” (30). This conclusion may be premature.
Dramatic benefits seen in small observational studies suggest larger RCTs might not be needed
Several clinical trials of experimental agents were undertaken during the Ebola outbreak in West Africa. Unlike statin/ARB treatment, all of the experimental agents tested targeted the Ebola virus, and none showed meaningful improvement in patient survival (28-30). In the largest trial (31), patients were treated consecutively and their outcomes were compared with those of historical controls. This is the same approach that was used in the vitamin C/corticosteroid and statin/ARB experiences described above (23,26,27). Moreover, when treatment benefits are five to ten times better than those of a comparable historical control group, an RCT might not be necessary (32). In such instances, it would be necessary to assume a preposterous level of confounding for the results not to indicate true benefit. It is important to keep in mind that benefits of this magnitude were seen in sepsis patients treated with high dose vitamin C and corticosteroids (23) and in Ebola patients treated with atorvastatin and irbesartan (2,26,27).
As noted above, the experiences of physicians who treated Ebola patients in Sierra Leone have either been ignored or dismissed as anecdotal. Virtually all investigators who planned, participated in or evaluated the trial experiences in West Africa favored the use of formal clinical trials, preferably RCTs (2,28-30). The same preference was expressed in a comprehensive report by a committee of the National Academies of Science, Engineering and Medicine in the US (33). The committee did, however, note “special situations” in which an uncontrolled phase 3 trial might be justified. This conclusion was based on the views of statisticians who in 1990 considered circumstances in which a formal clinical trial of an AIDS treatment might not be appropriate (Table 1) (33,34). All of the circumstances that could be used to justify an uncontrolled clinical trial were met for the statin/ARB Ebola treatment regimen, either initially or retrospectively when the results of treatment became known.
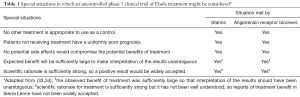
Full table
Combination treatment might be better than single agent treatment
In addition to demonstrating the value of serial clinical observations, the vitamin C/corticosteroid and statin/ARB experiences illustrate the additive or synergistic benefits of treating patients with combinations of two (or more) agents, a suggestion that was made almost a decade ago (35). Single drug treatment might be a reason for statin failure seen in some observational studies and in the RCTs conducted in sepsis/ARDS patients.
A recent report has confirmed the value of combination treatment in patients with community-acquired pneumonia. In a large retrospective cohort study, the reduction in 30-day all-cause mortality in those who received outpatient treatment with both a statin and an ARB was almost twice that of those treated with only one of these drugs (2,14,36). Combination treatment with statins and ARBs has a solid scientific rationale (2,37) and the two drugs have been used together in clinical practice for many years. The statin/ARB combination has one important advantage over high dose vitamin C/corticosteroid treatment: it does not require intravenous administration of either drug. It is even possible that combining a statin with a steroid might overcome the disadvantages of steroid treatment alone (38).
Combination treatment might reduce mortality due to pandemic influenza and other emerging virus diseases
For many years, physicians and health officials have been concerned about the possibility of a highly lethal pandemic caused by an avian influenza A (H5N1) virus (39). More recently, they have become concerned about the pandemic potential of influenza A (H7N9) viruses (40). Influenza virologists recognize the very real possibility that one of these viruses could combine with a seasonal human influenza virus and yield a reassortant that could cause a global pandemic (41). If and when this occurs, there is little likelihood that any country could obtain timely supplies of pandemic vaccines.
The shortcomings of relying solely on vaccination to confront a pandemic were made abundantly clear in 2009. Although the new influenza A (H1N1) pdm09 virus was not highly virulent, meaningful supplies of pandemic vaccines were unavailable in all countries during the first six months (42). In the US, a country better prepared than most, vaccination affected only 2–4% of all influenza cases, hospitalizations and deaths, and more than 90% of the world’s people had no timely access to pandemic vaccines and antivirals (2,42). When vaccines did become available, they arrived too late to do much good, and millions of doses went unused and had to be destroyed. Since then, the global capacity to produce seasonal and pandemic vaccines has greatly increased, but there has been no comparable increase in the global use of seasonal influenza vaccines, something regarded as essential for an effective vaccination response to the next pandemic (43). Chinese physicians and health officials are undoubtedly aware of these facts.
At a meeting organized by the Institute (now National Academy) of Medicine in 2004, I suggested that statins might be used for treatment and prophylaxis of pandemic influenza (44). This could be especially important for developing countries, which will have little hope of obtaining timely supplies of pandemic vaccines (45,46). However, inexpensive generic drugs like statins would be readily available on the first pandemic day. These drugs might also provide the means to mitigate the impact of an H7N9 pandemic (47). Unfortunately, the global community of influenza scientists and health officials who are concerned with pandemic preparedness have ignored (and continue to ignore) the possibility that these drugs might be the only way to reduce global mortality during the next pandemic (48). Perhaps the financial and reputational costs of adopting a new way of thinking about this are too great (49).
Evolution provides the conceptual foundation for treating the host response
In 1973, the geneticist Theodosius Dobzhansky wrote, “Nothing in biology makes sense except in the light of evolution” (50). Evolution provides the conceptual foundation for treating the host response to pandemic influenza and other emerging virus diseases (42,51).
In 2007, scientists wrote that during the 1918 influenza pandemic, “children were not protected from infection, but for reasons that are as mysterious today as they were in 1918, they were able to cope with the disease much better than their adult counterparts” (52). Almost a century later, this mortality difference has been replicated in pre- and post-pubertal mice (i.e., children and adults) (53). The molecular mechanisms that explain this mystery still await full discovery, but the broad outlines of our understanding have begun to emerge.
A recent report provides persuasive evidence that metabolic reprogramming is responsible for the anti-inflammatory effects of IL-10 in macrophages (54). This is one example of the remarkable acceleration of research into what has come to be called immunometabolism (55). This research may eventually explain many of the mechanisms responsible for both enhanced resistance to infection (the ability of the host to reduce the pathogen burden) and better tolerance of infection (the ability of the host to reduce its impact) (56).
More than a decade ago, clinical studies of human macrophages obtained from children and adults showed that children mount a much more anti-inflammatory response than adults to inflammatory stimuli (57,58). In the mouse model of influenza in children and adult mice described above, influenza virus loads in prepubertal mice (i.e., children) were essentially the same as virus loads in pubertal (i.e., adult) mice. Considered together, these findings suggest that children have a greater capacity to tolerate, not resist, influenza virus infection (2,42,51). It is likely that similar (if not the same) mechanisms explain the reduced mortality (i.e., better tolerance) of children to a variety of infectious diseases, including those caused by emerging viruses.
Almost ten years ago, investigators who were studying a highly inflammatory illness (hepatic ischemia reperfusion injury) in children and adult mice reported that when they treated the adults with an immunomodulatory drug (rosiglitazone), they changed the damaging inflammatory response of adults to the more benign (i.e., tolerant) response of children (59). In effect, they “rolled back” evolution (2,42,51). This finding suggests the very real possibility that immunomodulatory drugs might be used in the syndromic treatment of pandemic influenza and other emerging virus diseases (2,42).
Conclusions
The idea of treating the host response to pandemic influenza, Ebola and other emerging virus disease is based on four principles: (I) a Darwinian approach to therapeutic discovery grounded in observational studies, not the findings of incremental science and systems biology; (II) the idea that tolerance, not resistance, can be a defense strategy against infectious diseases, especially in children; (III) the mortality difference in children compared with adults seen in the 1918 influenza pandemic and in other infectious diseases has been hardwired by evolution; and (IV) treating the host response with widely available, inexpensive generic drugs can “roll back” evolution.
These four principles must be at the heart of all pandemic preparedness activities. Laboratory and clinical investigators still need to provide convincing evidence that treating the host response will improve survival. Responsibility for this research must be shared by investigators in all countries, not just those in the developed world. The reason for this is obvious; the mortality burden of these diseases will affect people everywhere.
Acknowledgements
None.
Footnote
Conflicts of Interest: The author has no conflicts of interest to declare.
References
- Zhang Z. Potential application of electronic healthcare records to the effectiveness study of the treatment of emerging virus diseases. J Emerg Crit Care Med 2017. [Epub ahead of print].
- Fedson DS. Treating emerging virus diseases: lessons learned from sepsis, pneumonia, influenza and Ebola. Ann Transl Med 2016;4:421. [Crossref] [PubMed]
- Mansur A, Steinau M, Popov AF, et al. Impact of statin therapy on mortality in patients with sepsis-associated acute respiratory distress syndrome (ARDS) depends on ARDS severity: a prospective cohort study. BMC Med 2015;13:128. [Crossref] [PubMed]
- Wan YD, Sun TW, Kan QC, et al. Effect of statin therapy on mortality from infection and sepsis: a meta-analysis of randomized and observational studies. Crit Care 2014;18:R71. [Crossref] [PubMed]
- Thomas G, Hraiech S, Loundou A, et al. Statin therapy in critically-ill patients with severe sepsis: a review and meta-analysis of randomized clinical trials. Minerva Anestesiol 2015;81:921-30. [PubMed]
- Nagendran M, McAuley DF, Kruger PS, et al. Statin therapy for acute respiratory distress syndrome: an individual patient data meta-analysis of randomised clinical trials. Intensive Care Med 2017;43:663-71. [Crossref] [PubMed]
- Alhazzani W, Truwit J. Statins in patients with sepsis and ARDS: is it over? Yes. Intensive Care Med 2017;43:672-4. [Crossref] [PubMed]
- Kruger PS, Terblanche M. Statins in patients with sepsis and ARDS: is it over? No. Intensive Care Med 2017;43:675-6. [Crossref] [PubMed]
- McAuley D, Charles PE, Papazian L. Statins in patients with sepsis and ARDS: is it over? We are not sure. Intensive Care Med 2017;43:677-9. [Crossref] [PubMed]
- Fedson DS, Baldwin JA. Previous hospital care as a risk factor for pneumonia: implications for immunization with pneumococcal vaccine. JAMA 1982;248:1989-95. [Crossref] [PubMed]
- Fedson DS, Harward MP, Reid RA, et al. Hospital-based pneumococcal immunization: epidemiologic rationale from the Shenandoah study. JAMA 1990;264:1117-22. [Crossref] [PubMed]
- Fedson DS, Wajda A, Nicol JP, et al. Influenza vaccination rates and risks for influenza-associated hospital discharge and death in Manitoba. Ann Intern Med 1992;116:550-5. [Crossref] [PubMed]
- Fedson DS, Wajda A, Nicol JP, et al. Clinical effectiveness of influenza vaccination in Manitoba. JAMA 1993;270:1956-61. [Crossref] [PubMed]
- Mortensen EM, Nakashima B, Cornell J, et al. Population-based study of statins, angiotensin II receptor blockers, and angiotensin-converting enzyme inhibitors on pneumonia-related outcomes. Clin Infect Dis 2012;55:1466-73. [Crossref] [PubMed]
- Joshi R, Venkatesan S, Myles PR. A UK general practice population cohort study investigating the association between lipid lowering drugs and 30-day mortality following medically attended acute respiratory illness. Peer J 2016;4:e1902. [Crossref] [PubMed]
- Li H, Yang SG, Gu L, et al. Effect of low-to-moderate dose corticosteroids on mortality of hospitalized adolescents and adults with influenza A(H1N1)pdm09 viral pneumonia. Influenza Other Respir Viruses 2017. [Epub ahead of print].
- Vandenbroucke JP. Case reports in an evidence-based world. J R Soc Med 1999;92:159-63. [Crossref] [PubMed]
- Fedson DS. Vaccination effectiveness, unmeasured confounding, and immunomodulatory treatment. J Infect Dis 2014;209:1300-1. [Crossref] [PubMed]
- Black S, Nicolay U, Del Giudice G, et al. Influence of statins on influenza vaccine responses in elderly individuals. J Infect Dis 2016;213:1224-8. [Crossref] [PubMed]
- Omer SB, Phadke VK, Bednarczyk RA, et al. Impact of statins on influenza vaccine effectiveness against medically attended acute respiratory illness. J Infect Dis 2016;213:1216-23. [Crossref] [PubMed]
- McLean HQ, Chow BD, VanWormer JJ, et al. The effect of statin use on influenza vaccine effectiveness. J Infect Dis 2016;214:1150-8. [Crossref] [PubMed]
- Fedson DS. Statins, influenza vaccination and influenza. J Infect Dis 2016;215:484-5. [PubMed]
- Marik PE, Khangoora V, Rivera R, et al. Hydrocortisone, vitamin C and thiamine for the treatment of severe sepsis and septic shock: a retrospective before-after study. Chest 2016. [Epub ahead of print]. [Crossref] [PubMed]
- Oudemans-van HM, Elbers PW, Spoelstra-de-Man AM. How to give vitamin C a cautious but fair chance in severe sepsis. Chest 2017. [Epub ahead of print]. [Crossref]
- Fedson DS. A practical treatment for patients with Ebola virus disease. J Infect Dis 2015;211:661-2. [Crossref] [PubMed]
- Fedson DS, Jacobson JR, Rordam OM, et al. Treating the host response to Ebola virus disease with generic statins and angiotensin receptor blockers. mBio 2015;6:e00716. [Crossref] [PubMed]
- Fedson DS, Rordam OM. Treating Ebola patients: a “bottom up” approach using generic statins and angiotensin receptor blockers. Int J Infect Dis 2015;36:80-4. [Crossref] [PubMed]
- Hayden FG, Friede M, Bausch DG. Experimental therapies for Ebola virus disease: what have we learned? J Infect Dis 2017;215:167-70. [PubMed]
- Duraffour S, Malvy D, Sissoko D. How to treat Ebola virus infections? A lesson from the field. Curr Opin Virol 2017;24:9-15. [Crossref] [PubMed]
- Rojek A, Horby P, Dunning J. Insights from clinical research completed during the west Africa Ebola virus disease epidemic. Lancet Infect Dis 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Sissoko D, Laouenan C, Folkesson E, et al. Experimental treatment with favipiravir for Ebola virus disease (the JIKI Trial): A historically controlled, single-arm proof-of-concept trial in Guinea. PLoS Med 2016;13:e1001967. [Crossref] [PubMed]
- Glasziou P, Chalmers I, Rawlins M, et al. When are randomised trials unnecessary? Picking signal from noise. BMJ 2007;334:349-51. [Crossref] [PubMed]
- Keusch G, McAdam K, Cuff P, et al. Integrating clinical research into epidemic response: the Ebola experience. Washington DC: The National Academies Press, 2017.
- Byar DP, Schoenfeld DA, Green SB, et al. Design considerations for AIDS trials. N Engl J Med 1990;323:1343-8. [Crossref] [PubMed]
- Fedson DS. Confronting an influenza pandemic with inexpensive generic agents: can it be done? Lancet Infect Dis 2008;8:571-6. [Crossref] [PubMed]
- Mortensen EM, Pugh MJ, Anzueto A. Prior use of both a statin and ARB is associated with lower mortality for patients hospitalized with pneumonia. Abs. European Respiratory Society, London, 3-7 September 2016.
- Lee HY, Sakuma I, Ihm AH, et al. Statins and renin-angiotensin system inhibitor combination treatment to prevent cardiovascular disease. Circ J 2014;78:281-7. [Crossref] [PubMed]
- Maneechotesuwan K, Ekjiratrakul W, Kasetsinsombat K, et al. Statins enhance the anti-inflammatory effects of inhaled corticosteroids in asthmatic patients through increased induction of indoleamine 2, 3-dioxygenase. J Allergy Clin Immunol 2010;126:754-62.e1. [Crossref] [PubMed]
- Harfoot R, Webby RJ. H5 influenza, a global update. J Microbiol 2017;55:196-203. [Crossref] [PubMed]
- Huo X, Chen L, Qi X, et al. Significantly elevated number of human infections with H7N9 virus in Jiangsu in eastern China, October 2016 to January 2017. Euro Surveill 2017;22:30496. [Crossref] [PubMed]
- Abente EJ, Kitikoon P, Lager KM, et al. A highly pathogenic avian influenza virus H5N1 with 2009 pandemic H1N1 internal genes demonstrates increased replication and transmission in pigs. J Gen Virol 2017;98:18-30. [Crossref] [PubMed]
- Fedson DS. Treating influenza with statins and other immunomodulatory agents. Antiviral Res 2013;99:417-35. [Crossref] [PubMed]
- Palache A, Oriol-Mathieu V, Fino M, et al. Seasonal influenza vaccine dose distribution in 195 countries (2004-2013): Little progress in estimated global vaccination coverage. Vaccine 2015;33:5598-605. [Crossref] [PubMed]
- Fedson DS. Pandemic influenza: a potential role for statins in treatment and prophylaxis. Clin Infect Dis 2006;43:199-205. [Crossref] [PubMed]
- Fedson DS, Dunnill P. Commentary: From scarcity to abundance: pandemic vaccines and other agents for "have not" countries. J Public Health Policy 2007;28:322-40. [Crossref] [PubMed]
- Fedson DS. Meeting the challenge of influenza pandemic preparedness in developing countries. Emerg Infect Dis 2009;15:365-71. [Crossref] [PubMed]
- Opal SM, Fedson DS. The dysfunctional host response to influenza A H7N9: a potential treatment option? Crit Care 2014;18:135. [Crossref] [PubMed]
- Fedson DS. How will physicians respond to the next influenza pandemic? Clin Infect Dis 2014;58:233-7. [Crossref] [PubMed]
- Baddeley M. Herding, social influences and behavioural bias in scientific research. EMBO Rep 2015;16:902-5. [Crossref] [PubMed]
- Dobzhansky T. Nothing in biology makes sense except in the light of evolution. Am Biol Teacher 1973;35:125-9. [Crossref]
- Fedson DS. Confronting the next influenza pandemic with anti-inflammatory and immunomodulatory agents: why they are needed and how they might work. Influenza Other Respir Viruses 2009;3:129-42. [Crossref] [PubMed]
- Ahmed R, Oldstone MB, Palese P. Protective immunity and susceptibility to infectious diseases: lessons from the 1918 influenza pandemic. Nat Immunol 2007;8:1188-93. [Crossref] [PubMed]
- Suber F, Kobzik L. Modeling childhood resistance to influenza mortality: increased survival in pre-pubertal and delayed puberty mice. Am J Respir Crit Care Med 2013;187:A1704.
- Ip WK, Hoshi N, Shouval DS, et al. Anti-inflammatory effect of IL-10 mediated by metabolic reprogramming of macrophages. Science 2017;356:513-9. [Crossref] [PubMed]
- Norata GD, Caligiuri G, Chavakis T, et al. The cellular and molecular basis of translational immunometabolism. Immunity 2015;43:421-34. [Crossref] [PubMed]
- Medzhitov R, Schneider DS, Soares MP. Disease tolerance as a defense strategy. Science 2012;335:936-41. [Crossref] [PubMed]
- Barsness KA, Bensard DD, Partrick DA, et al. Endotoxin induces an exaggerated interleukin-10 response in peritoneal macrophages of children compared with adults. J Pediatr Surg 2004;39:912-5; discussion 912-5. [Crossref] [PubMed]
- Barsness KA, Bensard DD, Partrick DA, et al. IL-1beta induces an exaggerated pro- and anti-inflammatory response in peritoneal macrophages of children compared with adults. Pediatr Surg Int 2004;20:238-42. [Crossref] [PubMed]
- Shin T, Kuboki S, Huber N, et al. Activation of peroxisome proliferator-activated receptor-gamma during hepatic ischemia is age-dependent. J Surg Res 2008;147:200-5. [Crossref] [PubMed]
Cite this article as: Fedson DS. Observational studies help us understand how to treat pandemic influenza and other emerging virus diseases. J Emerg Crit Care Med 2017;1:10.


