
A rare case of fulminant GBS mimicking brain death with clinical recovery
Introduction
In 1916, Guillain, Barré and Strohl described the main clinical features of Guillain-Barré syndrome (GBS): motor weakness, areflexia, minor paresthesias and cerebrospinal fluid (CSF) albumino-cytological dissociation. The frequent finding of motor conduction blocks and reduced conduction velocities provide confirmation of widespread demyelination. Common subtypes of GBS are acute inflammatory demyelinating polyradiculoneuropathy, acute motor axonal neuropathy, acute motor-sensory axonal neuropathy and Miller Fisher syndrome.
Fulminant cases of GBS mimicking brain death have been reported in which a rapid clinical deterioration occurs over hours to days (1,2). This clinical presentation is very rare, disease diagnosis can be challenging and patient mortality rate is high.
Case presentation
A 28-year-old male patient presented to emergency department with complaints of acute onset diplopia, difficulty in walking and inability to hold objects in his hands since waking on the day of admission. There was history of acute diarrhoeal illness 1 week prior to the onset of symptoms. His neurological exam was consistent with acute flaccid quadriparesis with complete areflexia in all four extremities. There was marked restriction of extraocular muscle movement in all primary directions of gaze. He had bifacial weakness, dysarthria and nasal intonation of voice. He followed commands briskly in spite of limited movements of limbs, eyes and facial musculature. On day 2 of ICU stay, his neurological exam became consistent with clinical brain death as he developed comatose appearance, bilaterally fixed and dilated pupils, absence of corneal reflex and complete loss of extraocular and facial movements. He required mechanical ventilation within 24 h of admission. His MRI brain and spine did not show any parenchymal lesion. Nerve conduction studies showed decreased H amplitude bilaterally with conduction block in left ulnar and right radial nerve suggestive of early acute inflammatory demyelinating polyneuropathy. CSF study on admission showed no cells with protein 44.5 mg/dL. He was started on intravenous immunoglobulins 0.4 gm/kg for 5 days starting from day 1. Repeat nerve conduction studies on day 7 showed inexcitable peripheral nerves. Repeat CSF study showed 5 cells with protein 57.7 mg/dL (Table 1). Electroencephalographic (EEG) study demonstrated intact cortical function with patient in persistently awake state. ANA and vasculitis panel, antibody to Acetylcholine receptor, urine for porphyrins and toxicology screen were negative. ELISA for HIV, CSF VDRL and Lyme disease serology were negative. Antiganglioside antibodies were negative. The patient developed autonomic dysfunction in the form of labile blood pressure and “tachy-brady syndrome”. He was treated with IV pulse Methylprednisolone in view of complete absence of response to IVIG. Tracheostomy and PEG tube insertion were planned in view of persistent neurological deficit. On day 18 of admission the patient did not show any change in neurological status. His serum IgG levels were 1,782 [791–1,643]. Second cycle of intravenous immunoglobulins (0.4 gm/kg) for 5 days was administered starting day 19.
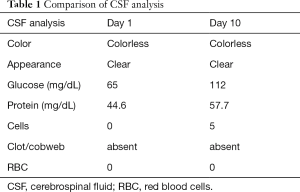
Full table
The patient remained clinically unresponsive for a period of 55 days during which he was provided DVT and bed sore prophylaxis, physiotherapy for prevention of contractures and muscle wasting and appropriate antibiotics for nosocomial infections. Plasma exchange (50 mL/kg) was carried out on 5 days from day 45 to 55 of hospital stay. The patient showed extraocular muscles and facial muscle movement from day 55 onwards. He showed progressive recovery over the period of next 45 days and was gradually weaned off mechanical ventilator support. He was discharged after 134 days of hospital stay (Table 2). At discharge, he was able to open and close eyes, move head to question /commands and able to mouth words. He had bladder and bowel control. At 9 months post onset of symptoms the patient is able to speak and swallow normally. MRC grading for muscle power at 9 months is as below.

Full table
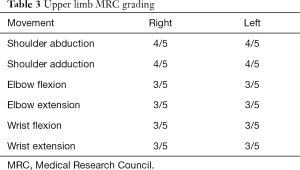
Full table
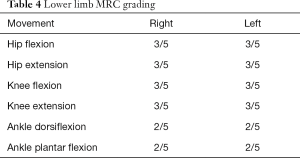
Full table
Discussion
GBS is the most common cause of acute flaccid paralysis worldwide with incidence of 1.8 per 100,000. There is slight male predominance with male: female ratio 1.5:1 (3). Dysautonomic features consisting of arrhythmias, hypertension or hypotension can occasionally turn fatal and are associated with poor clinical outcome (4). Mortality rate of 3 to 8 percent arises from complications like sepsis, acute respiratory distress syndrome (ARDS), pulmonary embolism or in rare cases, unexplained cardiac arrest, related to dysautonomia (3). Sixty-five percent of patients are left with persistent minor deficits, 20% with serious disability and only 20% with no residual problems (3). Fulminant GBS with absent braintem reflexes is a rare occurrence and is usually associated with high mortality. The diagnosis of fulminant GBS is based on rapidly progressive areflexic quadriparesis and early need for mechanical ventilation.
There is presence of albuminocytologic dissociation on CSF analysis with Nerve Conduction study suggestive of demyelination or primary axonal neuropathy (1). This condition has a poor recovery rate with high morbidity and mortality (5). Outcomes of this subset of patients have been described by Vargas et al. in the largest case series of 13 patients in 2000. Of the 3 deaths, 2 died from cardiac arrest due to dysautonomia and 1 died due to large anterior wall MI. Only 2 patients recovered with minor deficits and rest were severely disabled (5). Bernard et al. described a patient with fulminant GBS mimicking brain death who died from nosocomial infection after 158 days without any motor recovery (6). Nithyashree et al. described age above 60 years, prodromal diarrhoea, rapid involvement of the respiratory muscles requiring ventilatory support within 24 h of the illness, severe muscle weakness with grade 0 power in all four limbs, presence of cardiovascular autonomic symptoms and electrophysiology showing inexcitable peripheral nerves as factors indicating poor outcome in fulminant GBS (7). Our patient had all the above-mentioned features except that he was young. In general, the therapeutic response following IVIg is good in those with demyelinating neuropathy and unsatisfactory with axonal features (1). Few studies have shown that in those who are not responding to the first course of IVIg, a second course may show good response even in those with axonal changes (7,8). Our patient had received two cycles of intravenous immunoglobulins 2 weeks apart but remained unresponsive. The decision to treat with IVIg against Plasma exchange was taken considering severe dysautonomia though review of literature suggested equal efficacy of both modalities. However, when the patient did not show any response after two cycles of IVIg, Plasma exchange was done form day 45 to 55 of hospital stay. He responded well to the comprehensive care that included immunotherapy, rehabilitation and management of nosocomial infections. There has been progressive recovery of bulbar weakness and power in both upper and lower limbs. A review of literature for characteristics in cases of fulminant GBS with early requirement of mechanical ventilation can be summarized as below. All cases had requirement of mechanical ventilation within 48 h of admission. Absence of brainstem reflexes with coma is common to all cases. Neuroimaging was normal with nerve conduction study suggestive of severe demyelinating/axonal neuropathy (Table 5).
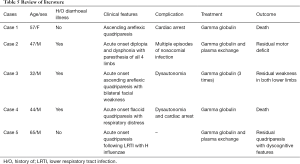
Full table
Conclusions
Fulminant GBS has a rapidly deteriorating clinical course associated with high mortality. Patients treated with multiple course of immunotherapeutic agents and persistent supportive care can have good outcome.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript.
References
- Hughes RA, Cornblath DR. Guillain-Barré syndrome. Lancet 2005;366:1653-66. [Crossref] [PubMed]
- Bernsen RA, de Jager AE, Schmitz PI, et al. Long-term impact on work and private life after Guillain-Barré syndrome. J Neurol Sci 2002;201:13-17. [Crossref] [PubMed]
- Yuki N, Hartung HP. Guillain-Barré syndrome. N Engl J Med 2012;366:2294-304. [Crossref] [PubMed]
- Patel MB, Goyal SK, Punnam SR, et al. Guillain-Barré Syndrome with asystole requiring permanent pacemaker: a case report. J Med Case Rep 2009;3:5. [Crossref] [PubMed]
- Vargas F, Hilbert G, Gruson D, et al. Fulminant Guillain-Barré syndrome mimicking cerebral death: case report and literature review. Intensive Care Med 2000;26:623-7. [Crossref] [PubMed]
- Bernard V, Van Pesch V, Hantson P. Guillain-Barré syndrome mimicking brain death pattern: a poorly reversible condition. Acta Neurol Belg 2010;110:93-6. [PubMed]
- Nithyashree N, Dhanaraj M, Kumar S, et al. Factors predicting poor outcome in patients with fulminant Guillaine-Barré syndrome. Ann Indian Acad Neurol 2014;17:463-5. [Crossref] [PubMed]
- Farcas P, Avnun L, Frisher S, et al. Efficacy of repeated intravenous immunoglobulin in severe unresponsive Guillain-Barré syndrome. Lancet 1997;350:1747. [Crossref] [PubMed]
Cite this article as: Trivedi T, Reddi R, Agarwal P. A rare case of fulminant GBS mimicking brain death with clinical recovery. J Emerg Crit Care Med 2017;1:20.


