Clinical scores for risk stratification of chest pain patients in the emergency department: an updated systematic review
Introduction
Chest pain is a common presenting symptom in the emergency department (ED). Many chest pain patients are admitted to the hospital due to the possibility of life threatening conditions, such as acute myocardial infarction (AMI) (1). It is however, not feasible to admit all chest pain patients due to limited healthcare resources (2). Therefore, distinguishing acute coronary syndrome (ACS) from other cardiac and non-cardiac diseases is crucial (3). It is essential to quickly and accurately identify patients who are at high and low risk of developing major adverse cardiac events (MACE) in order to optimally allocate ED and hospital resources.
Risk stratification of ED chest pain patients has been extensively studied in recent years (3). However, there is currently no widely accepted risk stratification method for ED chest pain patients (4). Initial ED risk scores were adopted from those created for post-ACS risk stratification such as the Thrombolysis in Myocardial Infarction (TIMI) score (5) and the Global Registry of Acute Coronary Events (GRACE) score (6), among others (7,8). However, because these risk scoring tools were not specifically designed for ED chest pain patients, their performance in the ED has been marginal (9-13).
The History, Electrocardiogram (ECG), Age, Risk factors, and initial Troponin (HEART) score was specifically created for risk stratifying ED patients with undifferentiated chest pain (14,15). Since its inception, there have been numerous validation studies to evaluate its effectiveness in diagnosing non-ST-elevation (NSTE) ACS (16,17). In many comparison studies, the HEART score was found to be superior to most existing risk stratification tools such as the TIMI and GRACE scores (4,18-21).
Because of growing patient censuses in many EDs, it is becoming increasingly important to quickly and accurately identify high risk chest pain patients to promote efficient and effective care. The purpose of this systematic review is to present an updated investigation on various risk stratification tools that are used in the ED to categorize chest pain patients according to their risk of developing MACE.
Methods
We searched PubMed and Embase, using keywords “(score OR scoring) AND (emergency department OR emergency room) AND (chest pain OR acute coronary syndrome)”. Our search was limited to English-language articles published between 01 January 2012 and 25 September 2017. Studies were eligible for inclusion if the reported clinical scores were used for risk stratifying ED chest pain patients, where one or more scoring methods were investigated. Systematic reviews, meta-analyses, case reports, letters to the editor, and articles without full text were excluded from this review. Studies with non-MACE outcomes were also excluded.
Initial literature search was conducted by N Liu. Two reviewers (N Liu and JC Ng) independently screened the titles, abstracts, and full articles for the inclusion of studies. Discrepancies were resolved through discussions among all authors to reach consensus. After screening for inclusion and exclusion criteria, data from the selected studies were extracted by N Liu. The extracted information included publication year, country of study, clinical scores investigated, sample size, study outcomes, summarized key information, and predictive performances including the area under the curve (AUC), sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). Since the aim of this systematic review was to present an updated summary of currently used risk stratification scores for ED chest pain patients, a meta-analysis was not conducted to rigorously compare the performances of various clinical scores.
Results
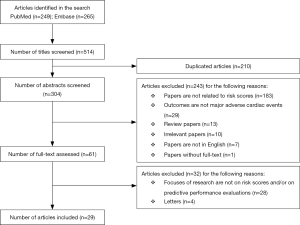
Our literature search identified a total of 514 articles, with 249 from PubMed database and 265 from Embase database. After removing 210 duplicated entries, we included 304 articles for abstract screening. We further excluded 243 articles due to reasons such as absence of risk stratification, non-MACE outcomes, etc. We reviewed the full-texts for the remaining 61 articles and further excluded 32 articles from the list; a final total of 29 articles were included in this systematic review (Figure 1).
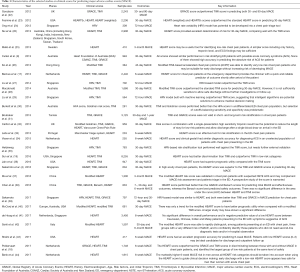
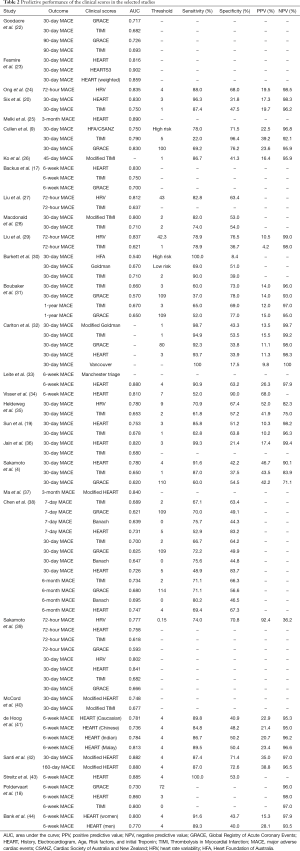
Characteristics of the 29 selected studies are summarized in Table 1. Predictive performances of the clinical scores in the selected studies are presented in Table 2. These studies were conducted in the Asia-Pacific (n=16) and Europe (n=11) and in more than 20 countries. Majority of the studies were conducted in Singapore (n=9), the Netherlands (n=5), the United States (n=5), and Australia (n=5). Four studies involved more than one country. Outcomes in all studies were MACE within a period after discharge from the ED, ranging from 72 hours to 1 year. Thirty-day MACE was the most commonly used outcome.
Full table
Full table
Seven out of 29 studies recruited more than 2,000 patients, with Sun et al.’s study (19) having the largest cohort of 8,255 patients obtained from the Internet Tracking Registry of Acute Coronary Syndromes (i*trACS) (45). More than 20 different risk scores or scoring methods were reported in these studies, which can be categorized into several main groups including the HEART score and its variants and the TIMI score and its variants. Among all scores, the HEART and the TIMI scores were the most widely validated; both scores appeared in 19 studies. The GRACE score and heart rate variability (HRV) based scores were also well studied.
TIMI score
The TIMI score was introduced in 2000 (5) and has since been widely adopted to assess the risk of MACE for patients with chest pain in the ED (46). There are seven elements in the TIMI score, namely age more than 65 years, more than three coronary artery disease (CAD) risk factors (hypertension, hyperlipidemia, diabetes, family history, and smoking), significant coronary artery stenosis, severe angina symptoms, ST-deviation, elevated cardiac enzymes, and use of aspirin in the last 7 days. Each element is assigned a score of 0 or 1. Therefore, the TIMI score is between 0 and 7.
Although not specifically designed for risk stratifying ED chest pain patients, the TIMI score and its variants have been widely applied and validated on the ED cohorts (46). In this systematic review, 19 out of 29 selected studies investigated TIMI based risk scores. The standard TIMI score has been implemented in most of the studies, and the modified TIMI score was also investigated in (26) and (28). In a study conducted in Australia, Macdonald et al. (28) validated a modified TIMI (mTIMI) score (range 0–10) (47) and found out that mTIMI outperformed standard TIMI score but was not sufficiently sensitive to allow safe discharge of low risk patients without further investigations.
Among the comparisons with various risk scores, the TIMI score achieved moderate performance in discriminating between chest pain patients with and without MACE (18-20). Furthermore, the TIMI score and its variants were reported as unreliable in identifying low risk chest pain patients (28,31).
GRACE score
Introduced in 2001, the GRACE score is a risk stratification tool developed based on the GRACE (6,48), studying an inpatient cohort rather than an ED cohort and initially designed to look at MACE outcomes at 6 months after hospital discharge (49). There are eight elements in the GRACE score, including age, heart rate, systolic blood pressure, creatinine, Killip class, cardiac arrest on admission, ST-deviation, and elevated cardiac enzymes.
External validations of the GRACE score showed moderate to poor performance in stratifying ED chest pain patients in distinguishing either the MACE group or the non-MACE group (17,18,31). However, the results are mixed. In the study by Cullen et al. (9), the GRACE score was found to be able to identify a sizable low risk cohort with high sensitivity and NPV. Yet, a study by Singer et al. (50) evaluating the frequency of missed AMIs in low-risk cohorts that were identified by established risk scores demonstrated that the GRACE score had the highest percentage of missed AMI, compared to the TIMI and HEART.
HEART score
Different from the TIMI and the GRACE scores, the HEART score was specifically developed for chest pain patients in the ED (14,20). It has been widely reported to outperform the TIMI and the GRACE scores (4,18,19). The HEART score has five prognostic factors, namely history, ECG, age, risk factors, and troponin. Each risk factor has a score of 0, 1, or 2, and the final score has a range of 0–10. The HEART score stratifies patients into three risk categories, that is, low risk [0–3], intermediate risk [4–6], and high risk [7–10]. It is noted that the HEART score was developed according to expert opinion (3), but not based on traditional multivariable regression analysis that is commonly used in clinical score derivation (51,52).
Due to its excellent performance in discriminating both high risk and low risk patients in terms of MACE development, the HEART score is the most validated score among all studied scores in this systematic review, where 19 out of 29 studies selected the HEART score as a main comparator. Comparing HEART with TIMI score in predicting 30-day MACE and 5-year all-cause mortality, Jain and colleagues (36) found that the HEART score was valuable in predicting not only short-term but also long-term outcomes for ED chest pain patients.
Other than the validations on general chest pain cohorts in the ED, the HEART score has been evaluated when several factors such as ethnicity and sex are taken into considerations. de Hoog et al. (41) conducted a study on a mixture of ethnic groups consisting of Caucasian, Chinese, Indian and Malay, and concluded that the overall performance of the HEART score was similar among Caucasians and Asians. Furthermore, Bank et al. (44) reported that male sex remained a significant factor for 6-week MACE when the HEART score was implemented for the prediction.
Based on the standard HEART score, many of its variants have been proposed and validated. Fesmire et al. proposed the HEARTS3 score by adding three additional variables, sex, serial 2-hour ECG, and serial 2-hour delta troponin (23). The HEARTS3 score reliably risk stratified patients with chest pain for MACE. Modified versions (37,40,42) of the HEART score were proposed where high-sensitivity troponin T was adopted to replace the traditional troponin element in the standard HEART score. These modifications suggested that the identified low-risk population might be directly discharged from the ED (40,42).
HRV score
HRV reflects the change in time intervals between heartbeats and has been shown to be a good predictor of MACE (53). Out of 29 studies selected in this review, Ong et al. (24), Liu et al. (29), and Sakamoto et al. (39) proposed several HRV-based risk scores to stratify ED chest pain patients with the purpose of making fast triage by predicting 72-hour MACE outcomes. Heldeweg et al. (35) developed a 30-day MACE prediction score SEDRSM by incorporating HRV, age, gender, and vitals through multivariable regression analysis. To calculate the score, all variables are discretized by proper thresholding and converted into individual scores. The range of SEDRSM is 0–37.
Liu et al. (29) and Liu et al. (27) used machine learning methods for variable selection and model derivation. In this review, most HRV-based scores were reported to outperform the TIMI score in predicting either 72-hour or 30-day MACE. Sakamoto et al. (39) made comparisons among the HRV score, the HEART score, the TIMI score and the GRACE score, and showed that the HRV score was superior to TIMI and GRACE scores, while achieving comparable performance with the HEART score. One advantage that the HRV-based scores have demonstrated over other clinical scores is their capability of computing the risk scores within a few minutes as only five-minute ECG records are needed for HRV parameter calculation (29).
Other scores
In addition to the above mentioned scores, several other clinical scores have been studied and evaluated, for example, the Vancouver chest pain rule (32), the Goldman risk score (30), the Heart Foundation of Australia (HFA)/Cardiac Society of Australia and New Zealand (CSANZ) guidelines (9), the Banach score (38), and the Manchester triage system (33). Cullen et al. (9) compared three risk scores (HFA/CSANZ, TIMI, and GRACE) and pointed out that all three scores had similar performance in predicting the risk of MACE for ED patients with chest pain. In Chen et al. (38), the Banach score had the largest AUC for predicting safety outcome while the HEART score had the largest AUC for predicting MACE.
Discussion
This systematic review provides an updated summary of studies on clinical scores for risk stratification of chest pain patients in the ED. The review was limited to articles published between 01 January 2012 and 25 September 2017. The initial literature search gave 514 published articles in the past 5 years that are related to risk stratification of ED chest pain patients. After screening and detailed verification according to inclusion and exclusion criteria, 29 articles were eventually selected for further review. More than 20 risk scores or scoring methods were found in these 29 studies and among them the HEART score and the TIMI score were the most investigated risk stratification tools. The selected studies have been well distributed among more than 20 countries, covering a mixture of diverse patient cohorts. Thirty-day and 6-week MACE were the most commonly used outcomes, and long-term outcomes followed up at 6-month (38,42) were also investigated.
As one of the most common reasons for emergency hospital admission, chest pain receives much attention as it is sometimes difficult to discern the etiology quickly and accurately (3). For efficient and accurate patient care, it is essential to develop strategies for rapid rule-out or rule-in of MACE. Most chest pain scores use troponin or other laboratory tests which require time. The pathway using high sensitivity cardiac troponin may be done in a hour, but still requires two blood tests (16). Although point-of-care (POC) cardiac biomarker testing have been gaining interest in recent years and have been developed to overcome the long turnaround time of laboratory testing, POC testing is still largely unavailable in most countries (54). Therefore, there seems to still be needs for faster tools to accurately risk stratify chest pain patients presenting to the ED. Over the years, many reviews have been published, ranging from general topics related to diagnosis of ACS (8,55,56) to systematic reviews on specific risk scores (16,46). Long and Koyfman have particularly studied current controversies in evaluating low risk chest pain patients with the aids of risk scores (1). Our systematic review aimed to provide a summary of the latest studies on risk scores for ED chest pain patient, therefore the focus was not on rigorous meta-analysis of predictive performance. Instead, we targeted at listing out widely used clinical scores and scoring methods in recent years, and summarizing their key characteristics.
The HEART, TIMI, and GRACE scores are three established stratification tools to assess the risk of MACE for chest pain patients. As a score that was specifically developed for chest pain patients in the ED, the HEART score is generally considered to outperform most other risk scores in discriminating high and low risk chest pain patients. This discovery has been consistent in most of the selected studies in this review, although some studies reported that there was no significant difference in terms of diagnostic accuracy between the HEART score and the TIMI score (38) or clinical gestalt (34). The HEART score was found particularly useful for identifying ED chest pain patients who had low probability of developing MACE (16,18). Marcoon et al. (57) proposed using the HEART score to further risk stratify patients with low TIMI scores and they suggested that a combined use of the HEART score and the TIMI score could identify a subgroup of patients with very low risk of developing MACE.
Other than conventional clinical scores, HRV-based scores and scoring methods were widely discussed (24,29,39) with the added value of faster risk stratification of chest pain patients. HRV techniques have been applied to develop a triage tool in the ED (39) where short-term outcome (i.e., 72-hour MACE) was adopted. With this tool, quicker response to high or low risk patients is possible as laboratory tests are not required. This may potentially shorten the process of risk prediction to a few minutes (29,35,39), compared to hours of waiting time in traditional chest pain pathways (58). However, the main disadvantage of HRV is its low interpretability. Ong et al. (24) and Heldeweg et al. (35) proposed risk scores by categorizing the HRV parameters through simple thresholding. Alternatively, Sakamoto et al. (39) and Liu et al. (29) used continuous HRV parameters for predictive modelling by either traditional logistic regression or advanced machine learning methods. Although the HRV-based scores are reported to achieve good performance compared to scores such as the TIMI and GRACE scores, they need extensive external validations. Future work may combine the strengths of the HRV technique and the clinical scores to create robust, fast, and accurate scores for stratifying chest pain patients in the ED. Machine learning (59) may also play important roles in selecting significant variables (27) and improving predictive performance (60).
Limitations
This systematic review has limitations. First, this review only included studies that were published in the past 5 years. Second, the focus of the review was on studies that reported score derivations and validations. It is noted that studies on clinical pathways using the risk scores were excluded. Lastly, meta-analysis was not conducted due to the heterogeneity of the study cohorts and the outcomes in the selected articles.
Conclusions
Chest pain, as a common yet potentially life threatening condition, deserves much attention in risk stratification and management, particularly in the ED where quick decisions are required for efficient patient care. This systematic review presents the latest studies on clinical scores that are used to categorize ED chest pain patients according to their risk of MACE. Traditional HEART, TIMI, and GRACE scores including their variants have been widely implemented and externally validated. Emerging techniques such as the HRV and machine learning were also adopted for developing risk scores where their flexibility and fast risk stratification were attractive, though low interpretability was a concern. In this review, risk score based clinical pathways were not discussed. Further investigations of existing risk scores including clinical outcomes and health economic measures are needed.
Acknowledgements
None.
Footnote
Conflicts of Interests: N Liu and ME Ong have a patent filing related to heart rate variability (system and method of determining a risk score for triage, application number: US 13/791,764); ME Ong has a similar patent filing unrelated to this study (method of predicting acute cardiopulmonary events and survivability of a patient, application number: US 13/047,348) and he also has a licensing agreement with ZOLL Medical Corporation for the above patented technology. The other authors have no conflicts of interest to declare.
References
- Long B, Koyfman A. Best Clinical Practice: Current Controversies in the Evaluation of Low-Risk Chest Pain with Risk Stratification Aids. Part 2. J Emerg Med 2017;52:43-51. [Crossref] [PubMed]
- Lee TH, Goldman L. Evaluation of the patient with acute chest pain. N Engl J Med 2000;342:1187-95. [Crossref] [PubMed]
- Backus BE, Six AJ, Kelder JH, et al. Risk scores for patients with chest pain: evaluation in the emergency department. Curr Cardiol Rev 2011;7:2-8. [Crossref] [PubMed]
- Sakamoto JT, Liu N, Koh ZX, et al. Comparing HEART, TIMI, and GRACE scores for prediction of 30-day major adverse cardiac events in high acuity chest pain patients in the emergency department. Int J Cardiol 2016;221:759-64. [Crossref] [PubMed]
- Antman EM, Cohen M, Bernink PJ, et al. The TIMI risk score for unstable angina/non-ST elevation MI: A method for prognostication and therapeutic decision making. JAMA 2000;284:835-42. [Crossref] [PubMed]
- Granger CB, Goldberg RJ, Dabbous O, et al. Predictors of hospital mortality in the global registry of acute coronary events. Arch Intern Med 2003;163:2345-53. [Crossref] [PubMed]
- Hollander JE, Than M, Mueller C. State-of-the-Art Evaluation of Emergency Department Patients Presenting With Potential Acute Coronary Syndromes. Circulation 2016;134:547-64. [Crossref] [PubMed]
- Fanaroff AC, Rymer JA, Goldstein SA, et al. Does This Patient With Chest Pain Have Acute Coronary Syndrome?: The Rational Clinical Examination Systematic Review. JAMA 2015;314:1955-65. [Crossref] [PubMed]
- Cullen L, Greenslade J, Hammett CJ, et al. Comparison of three risk stratification rules for predicting patients with acute coronary syndrome presenting to an Australian emergency department. Heart Lung Circ 2013;22:844-51. [Crossref] [PubMed]
- Chase M, Robey JL, Zogby KE, et al. Prospective validation of the Thrombolysis in Myocardial Infarction Risk Score in the emergency department chest pain population. Ann Emerg Med 2006;48:252-9. [Crossref] [PubMed]
- Ramsay G, Podogrodzka M, McClure C, et al. Risk prediction in patients presenting with suspected cardiac pain: the GRACE and TIMI risk scores versus clinical evaluation. QJM 2007;100:11-8. [Crossref] [PubMed]
- Lyon R, Morris AC, Caesar D, et al. Chest pain presenting to the Emergency Department--to stratify risk with GRACE or TIMI? Resuscitation 2007;74:90-3. [Crossref] [PubMed]
- Graham CA, Chan JW, Chan CP, et al. Prospective validation of Thrombolysis in Myocardial Infarction and front door Thrombolysis in Myocardial Infarction risk scores in Chinese patients presenting to the ED with chest pain. Am J Emerg Med 2014;32:1339-44. [Crossref] [PubMed]
- Six AJ, Backus BE, Kelder JC. Chest pain in the emergency room: value of the HEART score. Neth Heart J 2008;16:191-6. [Crossref] [PubMed]
- Backus BE, Six AJ, Kelder JC, et al. Chest pain in the emergency room: a multicenter validation of the HEART Score. Crit Pathw Cardiol 2010;9:164-9. [Crossref] [PubMed]
- Van Den Berg P, Body R. The HEART score for early rule out of acute coronary syndromes in the emergency department: a systematic review and meta-analysis. Eur Heart J Acute Cardiovasc Care 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Backus BE, Six AJ, Kelder JC, et al. A prospective validation of the HEART score for chest pain patients at the emergency department. Int J Cardiol 2013;168:2153-8. [Crossref] [PubMed]
- Poldervaart JM, Langedijk M, Backus BE, et al. Comparison of the GRACE, HEART and TIMI score to predict major adverse cardiac events in chest pain patients at the emergency department. Int J Cardiol 2017;227:656-61. [Crossref] [PubMed]
- Sun BC, Laurie A, Fu R, et al. Comparison of the HEART and TIMI Risk Scores for Suspected Acute Coronary Syndrome in the Emergency Department. Crit Pathw Cardiol 2016;15:1-5. [Crossref] [PubMed]
- Six AJ, Cullen L, Backus BE, et al. The HEART score for the assessment of patients with chest pain in the emergency department: a multinational validation study. Crit Pathw Cardiol 2013;12:121-6. [Crossref] [PubMed]
- Mahler SA, Hiestand BC, Goff DC Jr, et al. Can the HEART score safely reduce stress testing and cardiac imaging in patients at low risk for major adverse cardiac events? Crit Pathw Cardiol 2011;10:128-33. [Crossref] [PubMed]
- Goodacre SW, Bradburn M, Mohamed A, et al. Evaluation of Global Registry of Acute Cardiac Events and Thrombolysis in Myocardial Infarction scores in patients with suspected acute coronary syndrome. Am J Emerg Med 2012;30:37-44. [Crossref] [PubMed]
- Fesmire FM, Martin EJ, Cao Y, et al. Improving risk stratification in patients with chest pain: the Erlanger HEARTS3 score. Am J Emerg Med 2012;30:1829-37. [Crossref] [PubMed]
- Ong ME, Goh K, Fook-Chong S, et al. Heart rate variability risk score for prediction of acute cardiac complications in ED patients with chest pain. Am J Emerg Med 2013;31:1201-7. [Crossref] [PubMed]
- Melki D, Jernberg T. HEART score: a simple and useful tool that may lower the proportion of chest pain patients who are admitted. Crit Pathw Cardiol 2013;12:127-31. [Crossref] [PubMed]
- Ko H, Lee H, Ho H. A 2-hour Accelerated Chest Pain Protocol to Assess Patients with Chest Pain Symptoms in an Accident and Emergency Department in Hong Kong. Hong Kong Journal of Emergency Medicine 2013;20:261-9. [Crossref]
- Liu N, Koh ZX, Goh J, et al. Prediction of adverse cardiac events in emergency department patients with chest pain using machine learning for variable selection. BMC Med Inform Decis Mak 2014;14:75. [Crossref] [PubMed]
- Macdonald SP, Nagree Y, Fatovich DM, et al. Modified TIMI risk score cannot be used to identify low-risk chest pain in the emergency department: a multicentre validation study. Emerg Med J 2014;31:281-5. [Crossref] [PubMed]
- Liu N, Koh ZX, Chua EC, et al. Risk scoring for prediction of acute cardiac complications from imbalanced clinical data. IEEE J Biomed Health Inform 2014;18:1894-902. [Crossref] [PubMed]
- Burkett E, Marwick T, Thom O, et al. A comparative analysis of risk stratification tools for emergency department patients with chest pain. Int J Emerg Med 2014;7:10. [Crossref] [PubMed]
- Boubaker H, Beltaief K, Grissa MH, et al. Inaccuracy of Thrombolysis in Myocardial Infarction and Global Registry in Acute Coronary Events scores in predicting outcome in ED patients with potential ischemic chest pain. Am J Emerg Med 2015;33:1209-12. [Crossref] [PubMed]
- Carlton EW, Khattab A, Greaves K. Identifying Patients Suitable for Discharge After a Single-Presentation High-Sensitivity Troponin Result: A Comparison of Five Established Risk Scores and Two High-Sensitivity Assays. Ann Emerg Med 2015;66:635-45.e1. [Crossref] [PubMed]
- Leite L, Baptista R, Leitao J, et al. Chest pain in the emergency department: risk stratification with Manchester triage system and HEART score. BMC Cardiovasc Disord 2015;15:48. [Crossref] [PubMed]
- Visser A, Wolthuis A, Breedveld R, et al. HEART score and clinical gestalt have similar diagnostic accuracy for diagnosing ACS in an unselected population of patients with chest pain presenting in the ED. Emerg Med J 2015;32:595-600. [Crossref] [PubMed]
- Heldeweg ML, Liu N, Koh ZX, et al. A novel cardiovascular risk stratification model incorporating ECG and heart rate variability for patients presenting to the emergency department with chest pain. Crit Care 2016;20:179. [Crossref] [PubMed]
- Jain T, Nowak R, Hudson M, et al. Short- and Long-Term Prognostic Utility of the HEART Score in Patients Evaluated in the Emergency Department for Possible Acute Coronary Syndrome. Crit Pathw Cardiol 2016;15:40-5. [Crossref] [PubMed]
- Ma CP, Wang X, Wang QS, et al. A modified HEART risk score in chest pain patients with suspected non-ST-segment elevation acute coronary syndrome. J Geriatr Cardiol 2016;13:64-9. [PubMed]
- Chen XH, Jiang HL, Li YM, et al. Prognostic values of 4 risk scores in Chinese patients with chest pain: Prospective 2-centre cohort study. Medicine (Baltimore) 2016;95:e4778. [Crossref] [PubMed]
- Sakamoto JT, Liu N, Koh ZX, et al. Integrating heart rate variability, vital signs, electrocardiogram, and troponin to triage chest pain patients in the ED. Am J Emerg Med 2017. [Epub ahead of print]. [Crossref] [PubMed]
- McCord J, Cabrera R, Lindahl B, et al. Prognostic Utility of a Modified HEART Score in Chest Pain Patients in the Emergency Department. Circ Cardiovasc Qual Outcomes 2017;10:e003101. [Crossref] [PubMed]
- de Hoog VC, Lim SH, Bank IE, et al. HEART score performance in Asian and Caucasian patients presenting to the emergency department with suspected acute coronary syndrome. Eur Heart J Acute Cardiovasc Care 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Santi L, Farina G, Gramenzi A, et al. The HEART score with high-sensitive troponin T at presentation: ruling out patients with chest pain in the emergency room. Intern Emerg Med 2017;12:357-64. [Crossref] [PubMed]
- Streitz MJ, Oliver JJ, Hyams JM, et al. A retrospective external validation study of the HEART score among patients presenting to the emergency department with chest pain. Intern Emerg Med 2017. [Epub ahead of print]. [PubMed]
- Bank IE, de Hoog VC, de Kleijn DP, et al. Sex-Based Differences in the Performance of the HEART Score in Patients Presenting to the Emergency Department With Acute Chest Pain. J Am Heart Assoc 2017;6:e005373. [Crossref] [PubMed]
- Lindsell CJ, Anantharaman V, Diercks D, et al. The Internet Tracking Registry of Acute Coronary Syndromes (i*trACS): a multicenter registry of patients with suspicion of acute coronary syndromes reported using the standardized reporting guidelines for emergency department chest pain studies. Ann Emerg Med 2006;48:666-77, 677.e1-9.
- Hess EP, Agarwal D, Chandra S, et al. Diagnostic accuracy of the TIMI risk score in patients with chest pain in the emergency department: a meta-analysis. CMAJ 2010;182:1039-44. [Crossref] [PubMed]
- Body R, Carley S, McDowell G, et al. Can a modified thrombolysis in myocardial infarction risk score outperform the original for risk stratifying emergency department patients with chest pain? Emerg Med J 2009;26:95-9. [Crossref] [PubMed]
- Fox KA, Dabbous OH, Goldberg RJ, et al. Prediction of risk of death and myocardial infarction in the six months after presentation with acute coronary syndrome: prospective multinational observational study (GRACE). BMJ 2006;333:1091. [Crossref] [PubMed]
- GRACE Investigators. Rationale and design of the GRACE (Global Registry of Acute Coronary Events) Project: a multinational registry of patients hospitalized with acute coronary syndromes. Am Heart J 2001;141:190-9. [Crossref] [PubMed]
- Singer AJ, Than MP, Smith S, et al. Missed myocardial infarctions in ED patients prospectively categorized as low risk by established risk scores. Am J Emerg Med 2017;35:704-9. [Crossref] [PubMed]
- Adams ST, Leveson SH. Clinical prediction rules. BMJ 2012;344:d8312. [Crossref] [PubMed]
- Zhang Z, Zhang H, Khanal MK. Development of scoring system for risk stratification in clinical medicine: a step-by-step tutorial. Ann Transl Med 2017;5:436. [Crossref] [PubMed]
- Bigger JT, Fleiss JL, Rolnitzky LM, et al. The ability of several short-term measures of RR variability to predict mortality after myocardial infarction. Circulation 1993;88:927-34. [Crossref] [PubMed]
- Goodacre SW, Bradburn M, Cross E, et al. The Randomised Assessment of Treatment using Panel Assay of Cardiac Markers (RATPAC) trial: a randomised controlled trial of point-of-care cardiac markers in the emergency department. Heart 2011;97:190-6. [Crossref] [PubMed]
- Scirica BM. Acute coronary syndrome: emerging tools for diagnosis and risk assessment. J Am Coll Cardiol 2010;55:1403-15. [Crossref] [PubMed]
- Katus H, Ziegler A, Ekinci O, et al. Early diagnosis of acute coronary syndrome. Eur Heart J 2017;38:3049-55. [Crossref] [PubMed]
- Marcoon S, Chang AM, Lee B, et al. HEART score to further risk stratify patients with low TIMI scores. Crit Pathw Cardiol 2013;12:1-5. [Crossref] [PubMed]
- Long B, Oliver J, Streitz M, et al. An end-user's guide to the HEART score and pathway. Am J Emerg Med 2017;35:1350-5. [Crossref] [PubMed]
- Kononenko I. Machine learning for medical diagnosis: history, state of the art and perspective. Artif Intell Med 2001;23:89-109. [Crossref] [PubMed]
- Liu N, Sakamoto JT, Cao J, et al. Ensemble-Based Risk Scoring with Extreme Learning Machine for Prediction of Adverse Cardiac Events. Cognitive Computation 2017;9:545-54. [Crossref]
Cite this article as: Liu N, Ng JC, Ting CE, Sakamoto JT, Ho AF, Koh ZX, Pek PP, Lim SH, Ong ME. Clinical scores for risk stratification of chest pain patients in the emergency department: an updated systematic review. J Emerg Crit Care Med 2018;2:16.