
Paediatric acute respiratory distress syndrome: progress over the past decade
Introduction
Acute respiratory distress syndrome (ARDS) is lung disease characterised by severe diffuse inflammation and hypoxemia that can affect both adults and children. The cause can be of pulmonary or extra-pulmonary in origin. Sepsis is the most common risk factor in adults, whilst pneumonia in children (1,2). The reported incidence in children, termed paediatric ARDS (PARDS), is 2–12.8 per 100,000 person-years, which is much lower than that in adults (3,4). Outcome studies have revealed an overall mortality of 27–45% in adults with ARDS and 18–27% in children (4,5). However, mortality in adults and children have been decreasing in trend (6,7), probably owing to earlier recognition of the entity, better understanding of its pathophysiology and quicker instigation of appropriate treatment.
In this review, we focused on the following specific aspects of progress made in PARDS over the past decade: (I) the adoption of the new PARDS and neonatal ARDS definitions in clinical practice and research, in order to improve our understanding of ARDS epidemiology in the paediatric population; (II) novel biomarkers in risk stratification; (III) ventilatory strategies in PARDS, in particular the use of positive end-expiratory pressure (PEEP), tidal volumes and driving pressures; and (IV) personalized medicine approach in PARDS.
Methods
We used the following MeSH headings when searching for publications in PubMed: “positive end expiratory pressure” OR “biomarkers” OR “personalized medicine” AND “acute respiratory distress syndrome” OR “acute lung injury”. We limited our search to English publications and human studies in the last 10 years, but did not limit our search by publication type. We also hand-searched review and original articles on PARDS to include publications that were not captured in the initial search.
Evolution of definitions used in children with ARDS
Since the term Adult Respiratory Distress Syndrome was first coined in 1967 (8), and subsequently altered to ARDS due to the subsequent findings that this condition is confined to adults only. Since the first description, there has been various efforts made to redefine this syndrome, namely the American-European Consensus Conference (AECC) definition (9) and Berlin definition (10). The Paediatric Acute Lung Injury Consensus Conference (PALICC) in 2015 has been instrumental in providing more concise paediatric definitions for ARDS and to introduce comprehensive recommendations for treatment and future research focus (11). For the first time, PALICC proposed a definition of ARDS specific for children—the PARDS. Compared to the adult definition which requires the presence of bilateral chest infiltrates on radiograph, the PARDS definition recommends the presence of new infiltrate or infiltrates with an acute element attached, whether bilateral or not (Table 1). Apart from that, the paediatric definition utilized the oxygenation index or oxygenation saturation index as a marker of the degree of hypoxemia in these patients. These indices are commonly used in paediatric practice due to the potential challenges of gaining arterial access in children. Since the proposal of the PALICC’s definition of ARDS, there has been studies that examine the utility of this definition in prognostication of the disease process as well as to compare the robustness of this new definition against the adult definitions used in children. With standardized criteria for PARDS, a study of 4,764 children admitted to the intensive care unit demonstrated that the number of children diagnosed with ARDS increased (278 children under PALICC definition versus 134 children based on the AECC definition and 143 children as per Berlin definition) but overall mortality decreased (5.8% in the PALICC definition group versus 30.6% and 32.2% in the AECC and Berlin definition group, respectively) (13). In a multicenter, retrospective study based in Asia and involving 373 patients, mortality rate increased in a stepwise fashion according to the severity of PARDS as defined by PALICC, with overall mortality of 30.3% which is comparable to the AECC and Berlin definitions (14).
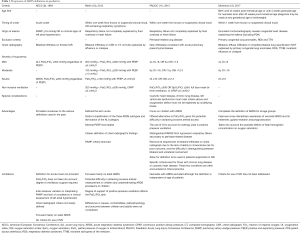
Full table
In the PALICC definition, the lower age limit was set to exclude patients with perinatal lung problems (11). It was soon after, in 2017 that this latter group was eventually included in the repertoire of ARDS definitions, as it was realised that the sequelae from acquired perinatal lung injuries such as meconium aspiration syndrome, were not that dissimilar to PARDS. In contrast to primary surfactant deficiency seen in neonatal respiratory distress syndrome, the concept of neonatal ARDS involve secondary dysfunction of surfactant or its amount with transient and minimal response to exogenous surfactant. Furthermore, to enhance cross-disciplinary knowledge, the Montreux definition for neonatal ARDS was established (12). We are currently awaiting more studies based on this new definition.
Progress have been made in the last two decades to improve the ARDS definition. The most obvious being the inclusion of the younger patients. Some of the advantages and limitations of all four definitions have been demonstrated in Table 1. We expect that these definitions will be updated regularly to reflect our improving understanding on the pathophysiology and management of children with PARDS.
Biomarkers
Many definitions exist for biomarkers, including one that has been led by the World Health Organization (WHO), “any substance, structure, or process that can be measured in the body or its products and influence or predict the incidence of outcome or disease” (15). For ARDS, biomarkers have been mainly obtained from blood, pulmonary tissues and bronchoalveolar lavage (BAL). Biomarkers from the breath of patients have also been used for analysis (16).
In the past few years, various efforts have been directed towards finding the one biomarker that could be used to identify those at risk of developing ARDS, to enable earlier detection and institution of treatment with subsequent evaluation of response to therapy, to determine prognosis and to guide the understanding of its pathophysiology. Biomarkers ought to ideally be accurate, reliable, cost-effective, and easily obtainable (16). They should be able to indicate the level of severity of illness with near precision, hence facilitating the type of treatment offered in time. The goal is to use biomarkers for bedside diagnosis, stratifying risk and molecular phenotyping to enable personalised treatment (17). Currently, multiple biomarkers have been described but none are convincingly reliable in their own right. A panel of biomarkers may be more feasible (18). In fact, sensitivity and specificity improves with a combination of biomarkers and clinical signs, as demonstrated by the enhancement in the area under the receiver operating characteristic (ROC) curve for predicting mortality when eight biomarkers (von Willebrand factor, surfactant protein D, tumour necrosis factor receptor-1, interleukin-6, interleukin-8, intercellular adhesion molecule-1, protein C and plasminogen activator inhibitor-1) were used together with clinical predictors in the ARDS Network trial in adults [area under the curve (AUC) for clinical predictors alone 0.815 (95% CI, 0.790–0.866); AUC for biomarkers alone 0.756 (95% CI, 0.733–0.821); AUC for clinical and biomarker predictors combined 0.850 (95% CI, 0.813–0.883)] (19). These biomarkers correspond to alveolar epithelial and capillary endothelial cell destruction, inflammation and clotting (20).
Many biomarkers investigated in paediatrics have been extrapolated from adult studies. This can be attributed mainly to the relative paucity of research in the area of PARDS due to the lower number and mortality rate of children with PARDS compared to adults with ARDS. A comprehensive overall review of biomarkers used in paediatric lung injury is available (21), and we refer the readers to this excellent review. Here we highlight selected biomarkers found mainly in adult studies that may have the potential to be analysed in PARDS, and other newer discoveries.
Receptor for advanced glycation end products (RAGE)
RAGE is a transmembrane protein receptor that can bind to multiple ligands. Its name stems from the initial finding of its binding to compounds from the glycol-oxidative reactions. However, it was noted later that other non-glycated peptides can also interact with the receptor. As one of the pattern-recognition receptors of the innate immune system, binding with its ligand triggers a signalling cascade which leads eventually to the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) transcription factor pathway cellular activation that induces generation of pro-inflammatory cytokines (22). RAGE is expressed at high levels in the lung, primarily by alveolar type I epithelial cells, responding rapidly to insults. However, ongoing inflammation would reverse this protective effect, leading to further injuries to the lung (22). The soluble form of RAGE (sRAGE) has been utilized in recent years as a biomarker of lung injury. Animal studies have shown an increase in these receptors upon induced-lung injury in blood and BAL. In a prospective study of 21 adults with ARDS, plasma sRAGE level peaked at day 1 and reduced over time. Those patients requiring more ventilatory support and oxygenation had significantly higher sRAGE readings in blood and BAL samples overall (23).
In another study of 20 patients with ARDS, sRAGE level was significantly higher in non-survivors compared to survivors (2,230.5±287.3 versus 1,796.8±418.5 pg/mL, P=0.002), and multivariate analysis revealed that sRAGE was independently associated with increased mortality in patients with ARDS (R2=0.289) (24). sRAGE has been noted as one of the biomarkers that strongly correlates with the diagnosis of ARDS in a high-risk population, according to a meta-analysis (25). There has only be a limited number of studies examining RAGE and sRAGE in PARDS. A prospective observational study in 80 children demonstrated a significant higher level of sRAGE in those with bronchiolitis compared to controls (1,215.7±535 versus 849±579 pg/mL, P<0.001) (26). In another study of 58 children aged less than 3 years old undergoing cardiac surgery, 16 (27.6%) of them developed acute lung injury after cardio-pulmonary bypass (CPB) with significantly elevated sRAGE level, even after 24 hours of surgery, compared to those without lung injury (one-way MANOVA: F=29.06; P<0.001 in immediately after CPB; F=11.72; P<0.001 in 24 hours after surgery). Multivariate analysis demonstrated that the sRAGE level immediately after CPB was an independent predictor for post-surgical acute lung injury (OR, 1.088; 95% CI, 1.011–1.171; P=0.025). Increased sRAGE level was also correlated with prolonged mechanical ventilation time, intensive care and hospital stay (27). There have been preliminary studies looking at the association between sRAGE levels in PARDS survivors and non-survivors and we look forward to the detailed results soon.
Angiopoietin-2 (Ang-2)
Another biomarker that has been known to be strongly associated with mortality from ARDS is Ang-2. This is an endothelial growth factor ligand released from endothelial cells and binds to tyrosine kinase receptor, resulting in increased vascular permeability and promoting inflammation (28).
In a prospective study of 230 adult patients, significantly higher levels of Ang-2 level were found in 19 patients who developed acute lung injury. By combining Ang-2 and lung injury prediction score, the area under ROC curve increased from 0.74 (95% CI, 0.62–0.84) using Ang-2 level alone or 0.74 (95% CI, 0.65–0.84) using lung injury prediction score alone, to 0.84 (95% CI, 0.78–0.91), improving the prediction of ARDS with the potential to initiate earlier treatment (29). In paediatrics, rising Ang-2 levels between days 1 and 3 of ARDS has been associated with increased mortality. In a cohort of 259 children with ARDS, a rising Ang-2 was shown in 61% of non-survivors compared with 42% of survivors (P=0.069). After adjusting for age, sex, race and PaO2/FiO2 ratio, the odds of mortality for a rising Ang-2 level were 3.3 (95% CI, 1.2–9.2, P<0.026). It was also revealed that those who survived the ARDS and had high Ang-2 level had increased morbidity as measured by the paediatric logistic organ dysfunction (PELOD) score (30).
Biomarkers in exhaled breath
An emerging exciting field in the ARDS world of biomarkers is the analysis of exhaled breath condensates and volatile organic compounds (VOCs) to predict the development of ARDS and to monitor progress. It is non-invasive, and with improved technology for sensing and algorithms for analysing, the technique of detection has been made possible recently (31). Constant analysis of airway gas samples via the ventilator by a mass spectrometry system can provide continuous readings of VOCs (32). A study of 53 adults in which 23 of them had ARDS demonstrated that the three breath metabolites: octane, acetaldehyde and 3-methylheptane, could discriminate between patients with and without ARDS. The area under the ROC curve was 0.8 (95% CI, 0.66–0.92), but improved to 0.91 (95% CI 0.85–0.97) when combined with lung injury prediction score (33). Till the best of our knowledge, there are no studies to date that examine exhaled breath in children with PARDS. Previous research in paediatrics have focused on the use of this technique to predict acute exacerbation of asthma based on six VOCs (34), and to discriminate between preschool children with and without wheeze according to 28 VOCs (35). Further studies in PARDS may be warranted to determine suitability, given the non-invasive nature of this method.
Nucleosome
Nucleosomes are histone/DNA complexes that circulate in the blood stream following cellular damage. They appear to be toxic to many different cell types. A prospective observational study was conducted in 76 children with PARDS. It was demonstrated that nucleosome levels were higher in those who did not survive lung injury compared to those who survived [0.59 AU (IQR 0.46–0.84) vs. 0.21 AU (IQR 0.08–0.33), rank sum P<0.001]. The level was also greater in those requiring more oxygenation support over the first 72 hours of disease, and is independently linked to increased mortality (36).
In summary, various biomarkers for ARDS exist and the idea is to determine the most appropriate ones to be used in a panel of tests, including with clinical markers to ensure accurate rapid early diagnosis. Currently, a prospective observational case-control study is ongoing to develop a biomarker-based early warning system of ARDS (37). Further research in paediatrics is required in this area.
Ventilatory strategies in PARDS
The use of PEEP
The use of lower tidal volume and higher PEEP termed as the lung-protective ventilation strategy has become the standard of care for ARDS over the last 2 decades (38,39). In the sixties when ARDS was first described, PEEP was mainly utilized to increase oxygenation. Over the years, PEEP is now regarded as a tool to avoid repetitive alveolar opening and closing, hence improve alveolar recruitment and reduce atelectrauma (40,41). However, clinicians must be mindful that adverse effects of excessive PEEP may outweigh the benefits. End-inspiratory alveolar over-distention associated with high PEEP worsens intrapulmonary shunt, pulmonary vascular resistance and increases alveolar dead space (42,43). The selection of an optimal PEEP level in the management of ARDS still remains controversial.
Due to lack of clinical trials in critically ill children with PARDS, the usage of tidal volume and PEEP in PARDS has been largely extrapolated from adult clinical trials (44). Earlier evidence that higher PEEP levels improve survival outcome was confined to animal models (45) and patients with severe ARDS (46). A cohort study by Gattinoni et al. later found that the effect of PEEP in ARDS was highly dependent on the percentage of recruitable lung (41). Higher PEEP levels aid in lung recruitment by keeping alveoli open throughout the ventilation cycle while lower PEEP levels, in contrast, propagate further lung injury as suggested by refractory hypoxemia (47). Overall, the usage of high PEEP level and low tidal volume has been termed lung-protective ventilation strategy and remains the mainstay ventilation technique of ARDS (48). This ventilation strategy is also adopted in the management of PARDS as shown in a retrospective study by Albuali et al. (49). However, the clinical benefit of high PEEP in children remains to be firmly demonstrated. In the prior-mentioned retrospective paediatric study of 164 children, investigators compared changes in mechanical ventilation for children with ARDS between 2 periods (49). Mean tidal volumes were lower and mean PEEP values were higher in the patients from the period of 2000 to 2004 compared to patients from 1988 to 1992. Survival rate was significantly better in patients from the later period. The authors found a significant association between decreased tidal volume and survival but were unable to demonstrate a significant association between PEEP levels and survival on multivariate analysis. The retrospective nature of this study makes it difficult to determine cause-effect relationships and prognostic values. Consensus recommendations from PALICC strongly recommended the need of clinical trials to assess lung recruitment strategies in PARDS. The consensus conference group did recognise that PEEP levels of more than 15 mmHg might be necessary in severe PARDS but close monitoring of oxygen delivery, respiratory compliance and haemodynamic is highly recommended (11).
The application of high PEEP levels was rationally thought to decrease mortality by decreasing repetitive alveolar opening and closing and hence reduce lung injury (50). A systematic review of high versus low PEEP levels in adult patients with ARDS (total patients =2,565) showed no statistical significance of barotrauma or ventilator free days between the two groups. Oxygenation on days 1, 3 and 7 was improved in the high PEEP group compared to the low PEEP group (44). In addition, a recent meta-analysis of eight randomised trials comparing high versus low PEEP levels in adult ARDS patients (total patients =2,728) also did not show any benefit of higher PEEP strategy in improving mortality, new organ failures or ventilator free days (51). An individual patient data meta-analysis (total patients =2,299) reported lower hospital mortality rate and higher rate of achieving unassisted breathing earlier in the higher PEEP group for patients with moderate-severe ARDS (PaO2/FiO2 ≤200) but not for patients with mild ARDS (52). The authors concluded that that use of higher PEEP was unlikely to improve clinical outcomes among unselected patients with ARDS. A recent multicentre randomised trial (total patients =1,013) compared low PEEP strategy with an experimental strategy of lung recruitment manoeuvre and PEEP titration. In this RCT, mean PEEP values throughout the 7 days trial were higher in the experimental group. The experimental group was not only associated with higher mortality at 28 days and 6 months, but also increased adverse effects such as barotrauma and haemodynamic instability (53). One major limitation of this RCT was the lack of blinding to both participants and clinicians, hence adding on to the possibility that care might be affected by treatment allocation.
Based on the various meta-analyses, high PEEP can improve oxygenation in ARDS even though it did not lead to significant reduction in mortality. The benefits of high PEEP levels are more apparent in patients with moderate to severe ARDS. A recent RCT, describe in the above paragraph, provided further clinical equipoise on the utility of PEEP in adults with ARDS, where higher mortality was demonstrated in patients supported with higher PEEP (53). One must be mindful that none of these meta-analyses and RCTs included children with PARDS. Whether high PEEP offers the same clinical benefit in children remains to be determined. To the best of our knowledge, there are currently no ongoing clinical trials on PARDS. Future RCTs are urgently needed to examine the impact of PEEP in children with PARDS.
Tidal volumes and driving pressures in PARDS
Over the last 20 years, there has been a shift of approach from high tidal volume to low tidal volume ventilation in ARDS. The benefits of low tidal volume strategy in improving survival in adults with ARDS was established in the landmark ARDS Network trial (54). Traditionally, high tidal volumes of 10 to 15 mL/kg of body weight were used to improve oxygenation and to achieve normal partial pressure of carbon dioxide and pH. Numerous studies have shown that the use of high tidal volumes was linked to disruption of pulmonary endothelium and release of inflammatory mediators, which eventually lead to propagation of the pro-inflammatory cascade and injury to the non-pulmonary organs (55,56). In the prior mentioned ARDS Network trial (total patients =861), mortality rate was reduced by 22% in the group receiving lower tidal volumes of 6 mL/kg body weight compared to the group receiving higher tidal volumes 12 mL/kg body weight. The lower tidal volume group also had significantly higher number of ventilation free-days and number of days without non-pulmonary organ failures (54). This landmark trial rebutted results of two earlier smaller RCTs (total patients =116 and 120 respectively) which showed no significant reduction of mortality rate in the lower tidal volume groups (7–8 mL/kg) compared to the higher tidal volume groups (10–12 mL/kg) (57,58). The discrepancies in findings can be attributed to the greater difference in tidal volume between groups and the larger number of patients in the ARDS landmark trial.
Similar to the titration of PEEP, the effects of tidal volume in PARDS remains unclear and largely inferred from adult studies. Contemporary practice of ventilation in PARDS does not involved very high tidal volumes as was used in the comparison group in the ARDS Network trial describe earlier (12 mL/kg). As such, a comparison trial between high and low tidal volumes in children with PARDS may be challenging to design and execute. Indeed, our experience of 373 children with PARDS across 10 PICUs in Asia did not show mortality differences in patients ventilated with tidal volumes between the range of 5 to 10 mL/kg (13). Careful design of future RCTs in the investigation of the impact of tidal volumes in PARDS is needed.
In recent years, there is growing interest in the investigation of the association between driving pressure to the outcomes of ARDS. Because the amount of “lung” available for ventilation is markedly decrease in ARDS, current recommendations of estimating tidal volume based on ideal body weight can generate different lung stress or strain (59,60). Driving pressure, measured as the airway pressure change from PEEP to end-inspiratory plateau pressure, is equivalent to the ratio between tidal volume and respiratory system compliance. Evidence has shown that driving pressure might be a better reflection of the functional lung size and hence better indicator of lung injury (59,61). A recent observational study of 3,562 patients found that airway driving pressure had stronger association with mortality compared to tidal volume and PEEP level; higher driving pressure was associated with increased mortality even when patients were receiving tidal volume in the “protective” range (62). The same study also found that the protective effects of higher PEEP was only observed when it was associated with a decreased driving pressure. As this study was an observation study, further RCTs are required to evaluate the effect of driving pressure in ARDS. Unfortunately, there also remains a lack of investigation in driving pressure in PARDS.
Personalised medicine
It has come to light fairly recently that many treatment trials for ARDS have failed to demonstrate clinical benefits in RCTs (e.g., corticosteroids, beta agonist, high PEEP) (63). A therapy that has been successful in preclinical studies or in a cohort of patients often is demonstrated futile in a larger population (64). One of the reasons for this could be the heterogeneity that exists amongst ARDS patients in terms of presentation, response to standard therapy and prognosis. The challenge would be to identify the appropriate subpopulation, preferably at the bedside, for the right treatment, ultimately reducing the probability of a trial showing no significant difference between the treatment and control groups. This area is rapidly developing in the adult world, but not so in PARDS, which needs to be addressed.
One of the ways to identify these subpopulations has been demonstrated in a study using the latent class analysis statistical method. Briefly, in this type of analysis, hidden groupings that is not directly measurable or observable in a dataset is uncovered. The observed data serves as an indicator to measure the concealed variables (65). Data from two previous RCTs in ARDS (52,54) were used to determine the subphenotypes of ARDS. One of the phenotypes identified had higher plasma concentrations of inflammatory biomarkers, severe shock, metabolic acidosis and significantly worse clinical outcome in terms of higher mortality, lesser ventilator-free days and organ-failure free days, compared to the other subphenotype. They also demonstrated differential response to PEEP in both subphenotypes. The 90-day mortality was 42% versus 51% for the hyper-inflammatory subphenotype when using higher versus lower PEEP, compared to 24% and 16% for the hypo-inflammatory subphenotype when using higher and lower PEEP, respectively (66).
Apart from that, the underlying aetiology of ARDS seems to play a role in causing the type of injuries succumbed by pulmonary tissues, namely epithelial injury if the ARDS insult was direct (e.g., pneumonia, aspiration), and endothelial injury in the situation of indirect ARDS (e.g., non-pulmonary sepsis). This can be inferred from the type of biomarkers raised. For example in direct ARDS, sRAGE will be increased compared to a rise in Ang-2 in indirect ARDS (67). The information is important if appropriate therapy is to be tailored towards the underlying cause.
Another area of development in personalized medicine is in the use of computer tomography of the chest to determine whether ARDS injury is focal or non-focal which would guide ventilatory strategies. According to this clinical trial, in those with focal ARDS, prone positioning will be promoted early, with use of moderate tidal volumes and low PEEP. In non-focal ARDS, recruitment manoeuvres, low tidal volume and high PEEP would be utilized. The aim is to decrease mortality at 90 days (68).
The Esophageal Pressure-Guided Ventilation 2 Trial is a multi-centre, RCT of ventilation in ARDS patients. This strategy uses oesophageal pressure to direct maintenance of minimal but positive transpulmonary pressure throughout ventilation (69). The pilot study revealed improvement in oxygenation and survival (70).
There is ongoing indication that ARDS should be managed with more precision given the heterogeneity nature of this condition. However, more research needs to be done, particularly in children.
Conclusions
In conclusion, the definition of ARDS has expanded beyond the original description to incorporate other populations that are also at risk for this disease (e.g., the neonatal population) With new definitions being introduced for ARDS in children (PARDS) and neonates (Montreux), we anticipate that future studies conducted in these groups of patients will increasingly utilized these definitions. The Montreux’s definition will require further validation before widespread adoption. The use of high PEEP in adults has been scrutinized recently but the overall impact of high PEEP in children with PARDS remains to be fully investigated. Various biomarkers of ARDS exist but efforts are directing towards establishing the most appropriate panel of investigations. In time to come, we expect that personalised medicine would improve management of individual or groups of patients with ARDS based on their unique underlying aetiology and manifestation.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Santschi M, Jouvet P, Leclerc F, et al. Acute lung injury in children: therapeutic practice and feasibility of international clinical trials. Pediatr Crit Care Med 2010;11:681-9. [Crossref] [PubMed]
- Rubenfeld GD, Caldwell E, Peabody E, et al. Incidence and Outcomes of Acute Lung Injury. N Engl J Med 2005;353:1685-93. [Crossref] [PubMed]
- Hough RF. Recent Advances in Pediatric Acute Respiratory Distress Syndrome (PARDS). Curr Pediatr Rep 2017;5:228-36. [Crossref]
- Schouten LRA, Veltkamp F, Bos AP, et al. Incidence and Mortality of Acute Respiratory Distress Syndrome in Children. Crit Care Med 2016;44:819-29. [PubMed]
- Khemani RG, Smith LS, Zimmerman JJ, et al. Pediatric Acute Respiratory Distress Syndrome. Pediatr Crit Care Med 2015;16:S23-40. [Crossref] [PubMed]
- Stapleton RD, Wang BM, Hudson LD, et al. Causes and timing of death in patients with ARDS. Chest 2005;128:525-32. [Crossref] [PubMed]
- Wong JJ, Jit M, Sultana R, et al. Mortality in Pediatric Acute Respiratory Distress Syndrome: A Systematic Review and Meta-Analysis. J Intensive Care Med 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Ashbaugh DG, Bigelow DB, Petty TL, et al. Acute respiratory distress in adults. Lancet 1967;2:319-23. [Crossref] [PubMed]
- Bernard GR, Artigas A, Brigham KL, et al. Report of the American-European consensus conference on ARDS: Definitions, mechanisms, relevant outcomes and clinical trial coordination. Intensive Care Med 1994;20:225-32. [Crossref] [PubMed]
- Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: The Berlin definition. JAMA 2012;307:2526-33. [PubMed]
- Pediatric Acute Lung Injury Consensus Conference Group. Pediatric Acute Respiratory Distress Syndrome: Consensus Recommendations from the Pediatric Acute Lung Injury Consensus Conference. Pediatr Crit Care Med 2015;16:428-39. [Crossref] [PubMed]
- De Luca D, van Kaam AH, Tingay DG, et al. The Montreux definition of neonatal ARDS: biological and clinical background behind the description of a new entity. Lancet Respir Med 2017;5:657-66. [Crossref] [PubMed]
- Parvathaneni K, Belani S, Leung D, et al. Evaluating the Performance of the Pediatric Acute Lung Injury Consensus Conference Definition of Acute Respiratory Distress Syndrome. Pediatr Crit Care Med 2017;18:17-25. [Crossref] [PubMed]
- Wong JJ, Phan HP, Phumeetham S, et al. Risk Stratification in Pediatric Acute Respiratory Distress Syndrome. Crit Care Med 2017;45:1820-8. [Crossref] [PubMed]
- Strimbu K, Tavel JA. What are biomarkers? Curr Opin HIV AIDS 2010;5:463-6. [Crossref] [PubMed]
- García-Laorden MI, Lorente JA, Flores C, et al. Biomarkers for the acute respiratory distress syndrome: how to make the diagnosis more precise. Ann Transl Med 2017;5:283. [Crossref] [PubMed]
- Ware LB, Calfee CS. Biomarkers of ARDS: what’s new? Intensive Care Med 2016;42:797-9. [Crossref] [PubMed]
- Ware LB, Koyama T, Zhao Z, et al. Biomarkers of lung epithelial injury and inflammation distinguish severe sepsis patients with acute respiratory distress syndrome. Crit Care 2013;17:R253. [Crossref] [PubMed]
- Ware LB, Koyama T, Billheimer DD, et al. Prognostic and pathogenetic value of combining clinical and biochemical indices in patients with acute lung injury. Chest 2010;137:288-96. [Crossref] [PubMed]
- Capelozzi VL, Allen TC, Beasley MB, et al. Molecular and Immune Biomarkers in Acute Respiratory Distress Syndrome: A Perspective From Members of the Pulmonary Pathology Society. Arch Pathol Lab Med 2017;141:1719-27. [Crossref] [PubMed]
- Orwoll BE, Sapru A. Biomarkers in Pediatric ARDS: Future Directions. Front Pediatr 2016;4:55. [Crossref] [PubMed]
- Guo WA, Knight PR, Raghavendran K. The receptor for advanced glycation end products and acute lung injury/acute respiratory distress syndrome. Intensive Care Med 2012;38:1588-98. [Crossref] [PubMed]
- Mauri T, Masson S, Pradella A, et al. Elevated plasma and alveolar levels of soluble receptor for advanced glycation endproducts are associated with severity of lung dysfunction in ARDS patients. Tohoku J Exp Med 2010;222:105-12. [Crossref] [PubMed]
- Nakamura T, Sato E, Fujiwara N, et al. Increased levels of soluble receptor for advanced glycation end products (sRAGE) and high mobility group box 1 (HMGB1) are associated with death in patients with acute respiratory distress syndrome. Clin Biochem 2011;44:601-4. [Crossref] [PubMed]
- Terpstra ML, Aman J, van Nieuw Amerongen GP, et al. Plasma Biomarkers for Acute Respiratory Distress Syndrome. Crit Care Med 2014;42:691-700. [Crossref] [PubMed]
- García-Salido A, Oñoro G, Melen GJ, et al. Serum sRAGE as a Potential Biomarker for Pediatric Bronchiolitis: A Pilot Study. Lung 2015;193:19-23. [Crossref] [PubMed]
- Liu X, Chen Q, Shi S, et al. Plasma sRAGE enables prediction of acute lung injury after cardiac surgery in children. Crit Care 2012;16:R91. [Crossref] [PubMed]
- Roviezzo F, Tsigkos S, Kotanidou A, et al. Angiopoietin-2 causes inflammation in vivo by promoting vascular leakage. J Pharmacol Exp Ther 2005;314:738-44. [Crossref] [PubMed]
- Agrawal A, Matthay MA, Kangelaris KN, et al. Plasma angiopoietin-2 predicts the onset of acute lung injury in critically ill patients. Am J Respir Crit Care Med 2013;187:736-42. [Crossref] [PubMed]
- Zinter MS, Spicer A, Orwoll BO, et al. Plasma angiopoietin-2 outperforms other markers of endothelial injury in prognosticating pediatric ARDS mortality. Am J Physiol Lung Cell Mol Physiol 2016;310:L224-31. [Crossref] [PubMed]
- Boots AW, Bos LD, van der Schee MP, et al. Exhaled Molecular Fingerprinting in Diagnosis and Monitoring: Validating Volatile Promises. Trends Mol Med 2015;21:633-44. [Crossref] [PubMed]
- Dolch ME, Frey L, Hornuss C, et al. Molecular breath-gas analysis by online mass spectrometry in mechanically ventilated patients: A new software-based method of CO2-controlled alveolar gas monitoring. J Breath Res 2008;2:037010. [Crossref] [PubMed]
- Bos LDJ, Weda H, Wang Y, et al. Exhaled breath metabolomics as a noninvasive diagnostic tool for acute respiratory distress syndrome. Eur Respir J 2014;44:188-97. [Crossref] [PubMed]
- Robroeks CM, Van Berkel JJ, Jöbsis Q, et al. Exhaled volatile organic compounds predict exacerbations of childhood asthma in a 1-year prospective study. Eur Respir J 2013;42:98-106. [Crossref] [PubMed]
- van de Kant KD, van Berkel JJ, Jöbsis Q, et al. Exhaled breath profiling in diagnosing wheezy preschool children. Eur Respir J 2013;41:183-8. [Crossref] [PubMed]
- Yehya N, Thomas NJ, Meyer NJ, et al. Circulating markers of endothelial and alveolar epithelial dysfunction are associated with mortality in pediatric acute respiratory distress syndrome. Intensive Care Med 2016;42:1137-45. [Crossref] [PubMed]
- Hu M. Establishment of a Biomarkers-based Early Warning System of Acute Respiratory Distress Syndrome (ARDS). ClinicalTrials.gov. (cited 14 Jan 2018). Available online: https://clinicaltrials.gov/ct2/show/NCT03236272
- Artigas A, Bernard GR, Carlet J, et al. Ventilatory, Pharmacologic, Supportive Therapy, Study Design Strategies, and Issues Related to Recovery and Remodeling and the Consensus Committee. Am J Respir Crit Care Med 1998;157:1332-47. [Crossref] [PubMed]
- Ranieri VM, Suter PM, Tortorella C, et al. Effect of Mechanical Ventilation on Inflammatory Mediators in Patients With Acute Respiratory Distress Syndrome A Randomized Controlled Trial. JAMA 1999;282:54-61. [Crossref] [PubMed]
- Gattinoni L, Pelosi P, Crotti S, et al. Effects of Positive End-Expiratory Pressure on Regional Distribution of Tidal Volume and Recruitment in Adult Respiratory Distress Syndrome. Am J Respir Crit Care Med 1995;151:1807-14. [Crossref] [PubMed]
- Gattinoni L, Caironi P, Cressoni M, et al. Lung Recruitment in Patients with the Acute Respiratory Distress Syndrome. N Engl J Med 2006;354:1775-86. [Crossref] [PubMed]
- Rouby JJ, Brochard L. Tidal recruitment and overinflation in acute respiratory distress syndrome: Yin and Yang. Am J Respir Crit Care Med 2007;175:104-6. [Crossref] [PubMed]
- Hubmayr RD. Perspective on lung injury and recruitment: A skeptical look at the opening and collapse story. Am J Respir Crit Care Med 2002;165:1647-53. [Crossref] [PubMed]
- Santa Cruz R, Rojas JI, Nervi R, et al. High versus low positive end-expiratory pressure (PEEP) levels for mechanically ventilated adult patients with acute lung injury and acute respiratory distress syndrome. Cochrane Database Syst Rev 2013.CD009098. [PubMed]
- Tremblay LN, Slutsky AS. Ventilator-induced lung injury: From the bench to the bedside. Intensive Care Med 2006;32:24-33. [Crossref] [PubMed]
- Amato MBP, Barbas CSV, Medeiros DM, et al. Effect of a Protective-Ventilation Strategy on Mortality in the Acute Respiratory Distress Syndrome. N Engl J Med 1998;338:347-54. [Crossref] [PubMed]
- Gattinoni L, Caironi P. Refining Ventilatory Treatment for Acute Lung Injury and Acute Respiratory Distress Syndrome. JAMA 2008;299:691-3. [Crossref] [PubMed]
- Petrucci N, De Feo C. Lung protective ventilation strategy for the acute respiratory distress syndrome. Cochrane Database Syst Rev 2013.CD003844. [PubMed]
- Albuali WH, Singh RN, Fraser DD, et al. Have changes in ventilation practice improved outcome in children with acute lung injury? Pediatr Crit Care Med 2007;8:324-30. [PubMed]
- Caironi P, Cressoni M, Chiumello D, et al. Lung opening and closing during ventilation of acute respiratory distress syndrome. Am J Respir Crit Care Med 2010;181:578-86. [Crossref] [PubMed]
- Walkey AJ, Del Sorbo L, Hodgson CL, et al. Higher PEEP versus Lower PEEP Strategies for Patients with Acute Respiratory Distress Syndrome. A Systematic Review and Meta-Analysis. Ann Am Thorac Soc. 2017;14:S297-303. [Crossref] [PubMed]
- Briel M, Meade M, Mercat A, et al. Higher vs Lower Positive End-Expiratory Pressure in Patients With Acute Lung Injury and Acute Respiratory Distress Syndrome Systematic Review and Meta-analysis. JAMA 2010;303:865-73. [Crossref] [PubMed]
- Cavalcanti AB, Suzumura ÉA, Laranjeira LN, et al. Effect of Lung Recruitment and Titrated Positive End-Expiratory Pressure (PEEP) vs Low PEEP on Mortality in Patients With Acute Respiratory Distress Syndrome. JAMA 2017;318:1335-45. [Crossref] [PubMed]
- Brower RG, Matthay MA, Morris A, et al. Ventilation with Lower Tidal Volumes as Compared with Traditional Tidal Volumes for Acute Lung Injury and the Acute Respiratory Distress Syndrome. N Engl J Med 2000;342:1301-8. [Crossref] [PubMed]
- Tsuno K, Miura K, Takeya M, et al. Histopathologic pulmonary changes from mechanical ventilation at high peak airway pressures. Am Rev Respir Dis 1991;143:1115-20. [Crossref] [PubMed]
- Slutsky AS, Tremblay LN. Multiple system organ failure. Is mechanical ventilation a contributing factor? Am J Respir Crit Care Med 1998;157:1721-5. [Crossref] [PubMed]
- Brochard L, Roudot-Thoraval F, Roupie E, et al. Tidal volume reduction for prevention of ventilator-induced lung injury in acute respiratory distress syndrome. The Multicenter Trail Group on Tidal Volume reduction in ARDS. Am J Respir Crit Care Med 1998;158:1831-8. [Crossref] [PubMed]
- Stewart TE, Meade MO, Cook DJ, et al. Evaluation of a ventilation strategy to prevent barotrauma in patients at high risk for acute respiratory distress syndrome. Pressure- and Volume-Limited Ventilation Strategy Group. N Engl J Med 1998;338:355-61. [Crossref] [PubMed]
- Gattinoni L, Pesenti A. The concept of "baby lung". Intensive Care Med 2005;31:776-84. [Crossref] [PubMed]
- Chiumello D, Carlesso E, Cadringher P, et al. Lung stress and strain during mechanical ventilation for acute respiratory distress syndrome. Am J Respir Crit Care Med 2008;178:346-55. [Crossref] [PubMed]
- Chiumello D, Carlesso E, Brioni M, et al. Airway driving pressure and lung stress in ARDS patients. Crit Care 2016;20:276. [Crossref] [PubMed]
- Amato MB, Meade MO, Slutsky AS, et al. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med 2015;372:747-55. [Crossref] [PubMed]
- Jabaudon M, Blondonnet R, Audard J, et al. Recent directions in personalised acute respiratory distress syndrome medicine. Anaesth Crit Care Pain Med 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Frank AJ, Thompson BT. Pharmacological treatments for acute respiratory distress syndrome. Curr Opin Crit Care 2010;16:62-8. [Crossref] [PubMed]
- Goodman LA. Exploratory latent structure analysis using both identifiable and unidentifiable models. Biometrika 1974;61:215-31. [Crossref]
- Calfee CS, Delucchi K, Parsons PE, et al. Subphenotypes in acute respiratory distress syndrome: Latent class analysis of data from two randomised controlled trials. Lancet Respir Med 2014;2:611-20. [Crossref] [PubMed]
- Calfee CS, Janz DR, Bernard GR, et al. Distinct molecular phenotypes of direct vs indirect ARDS in single-center and multicenter studies. Chest 2015;147:1539-48. [Crossref] [PubMed]
- Lung Imaging for Ventilatory Setting in ARDS. ClinicalTrials.gov. (cited 14 Jan 2018). Available online: https://clinicaltrials.gov/show/NCT02149589
- Fish E, Novack V, Banner-Goodspeed VM, et al. The Esophageal Pressure-Guided Ventilation 2 (EPVent2) trial protocol: a multicentre, randomised clinical trial of mechanical ventilation guided by transpulmonary pressure. BMJ Open 2014;4:e006356. [Crossref] [PubMed]
- Talmor D, Sarge T, Malhotra A, et al. Mechanical ventilation guided by esophageal pressure in acute lung injury. N Engl J Med 2008;359:2095-104. [Crossref] [PubMed]
Cite this article as: Wang J, Loh SW, Lee JH. Paediatric acute respiratory distress syndrome: progress over the past decade. J Emerg Crit Care Med 2018;2:24.


