
Current use and advances in vasopressors and inotropes support in shock
Introduction
Shock is a life-threatening, generalized state of circulatory failure resulting in the inability to deliver oxygen in peripheral tissues to meet their demands. Some clinical symptoms of shock include cold skin, increased capillary refill time (>3 seconds) and increased central-to-toe temperature gradient (1-3). More specifically, the state of shock is the result of one of the four following mechanisms. The first one is the decrease of the venous return due to the loss of the circulating volume (hemorrhagic shock) (3). The second one is the inability of the heart to function as a pump due to the loss of contractility or abnormal electrical activity such as arrhythmias (cardiogenic shock) (4). The third one is obstruction due to pulmonary embolism, tension pneumothorax and cardiac tamponade (obstructive shock) (1). The fourth one is loss of vascular tone due to maldistribution of blood (septic, anaphylactic and neurogenic shock) (5-7).
As far as the shocked patient is concerned, the earlier the diagnosis, the better the outcome; it is of crucial importance to identify early the presence of the state of shock (8). The skin (decreased tissue perfusion), the kidneys (decreased urine output) and the brain (impaired mental status) are the most easily accessible organs to assess the state of shock. It should be noted that the Expert Panel Recommendations on circulatory shock and hemodynamic monitoring suggest that the determination of the type of shock leads to the most appropriate therapeutic interventions, improving survival rates (1). The main priority in shock is to maintain the hemodynamics of the patient until the cause of shock can be identified. This is mainly achieved with fluid resuscitation, inotropic and vasopressor therapy (2,9).
The purpose of this review is to address both the current use of vasopressors and inotropes support in shock (thereby, offering a concise review of the pathophysiology behind shock alongside a helpful clinical reference tool for the emergency physician) as well as to describe recent advances (both experimental and clinical) that could hold a critical role for the near future regarding patient management.
Vasopressors and inotropes
Inotropes are a class of drugs that change the force of the heart’s contraction. The physiological basis for their actions, apart from its direct effect on cardiac myocyte excitation and contraction, is characterized by changes in the homeostasis of the microvascular flow, alterations of the metabolic rate through the production of metabolically active molecules and alterations in the state of activation of immune cells (10).
Vasopressors are a heterogeneous class of drugs with powerful and immediate hemodynamic effects that increase the tone of the vascular system (vasotonus) and, therefore, mean arterial pressure (MAP). Restoration of adequate pressure is the criterion of their effectiveness; however, blood pressure (BP) does not always equate to blood flow. The relative potency of various vasopressors on cardiac heart rate and contractility and peripheral vasculature tone depends on receptor distribution and their corresponding affinity for them. Vasopressors can be classified according to their adrenergic and non-adrenergic actions (10). Adrenergic agents include norepinephrine, phenylephrine, epinephrine, dopamine, dobutamine and isoproterenol. Non-adrenergic agents include angiotensin II and nitric oxide (NO) inhibitors. Many drugs have both vasopressor and inotropic effects (10).
Vasopressors and inotropes act on alpha adrenergic, beta adrenergic, dopamine, calcium sensitizers and angiotensin receptors (11,12). Alpha-1 adrenergic receptors are located in vascular walls and induce significant vasoconstriction. Alpha-1 adrenergic receptors are, also, located in the heart and can increase the duration of contraction without increasing chronotropy, that is the HR (13). Beta-2 adrenergic receptors in vessels induce vasodilation (11). Dopamine receptors are present in the cerebral, coronary, mesenteric and renal vascular beds. When stimulated, they induce vasodilation. Dopamine receptor subtypes are responsible for norepinephrine release which causes vasoconstriction (11). Some of these drugs increase the sensitivity of the myocardial contractile apparatus to calcium, leading to increase in inotropy and vasodilation (12,13).
Current knowledge
Cardiogenic shock
Cardiogenic shock is a state of impaired end-organ perfusion caused by a decrease in CO despite adequate intravascular volume and is, usually, associated with the following hemodynamic characteristics: systolic blood pressure (SBP) <90 mmHg for more than 30 min (in the absence of inotropic or vasopressor support), a reduction of cardiac index (<1.8 L/min/m2 without support and <2.2 L/min/m2 with support) and elevated left ventricular (LV) filling pressures (pulmonary capillary wedge pressure >18 mmHg) (14).
LV dysfunction is the most common cause of cardiogenic shock complicating Acute Myocardial Infarction (AMI). In general, a loss of >40% of functional myocardium is required to cause cardiogenic shock. Less frequent causes of cardiogenic shock secondary to AMI are mechanical complications, such as acquired ventricular septal defect, free wall rupture and papillary muscle rupture or dysfunction with subsequent acute ischemic mitral regurgitation. Furthermore, acute right ventricular infarction might cause cardiogenic shock (15).
The SHOCK trial, one of the most popular trials regarding cardiogenic shock, suggested that early revascularization in AMI patients (coronary artery bypass grafting, CABG or percutaneous coronary intervention, PCI), was accompanied by a lower rate of mortality when compared to the initial patient medical stabilization (4). This is the reason why revascularization is considered the definitive treatment of patients with shock after AMI. The mechanical augmentation of cardiac function with intra-aortic balloon pump or left ventricular assisted devices (LVADs) should only be used as a temporary solution until definitive revascularization treatment (16).
Hemodynamic alterations in cardiogenic shock complicating AMI or decompensated heart failure are addressed by the use of vasopressors and/or inotropes. Vasopressors are administered to raise BP and restore end-organ perfusion (17). Inotropes are used as they increase myocardial contractility, thereby, increasing CO. Often this kind of agents increase HR; subsequently, the increased myocardial oxygen consumption worsens the condition of the patient. Along with the positive inotropic properties, these drugs manifest peripheral vasoconstrictive and vasodilatory effects (18). In general, vasopressor agents and inotropes should be used in clinical practice at the lowest doses and the shortest period possible.
Dobutamine increases HR, stroke volume (SV) and CO with a concomitant decrease in left ventricular filling pressures (LVFP) and a modest decrease in BP and SVR, thus it seems to be the ideal inotropic agent in cardiogenic shock. However, data from the Acute Decompensated Heart Failure National Registry revealed an increase in mortality due to the manifested precipitating myocardial ischemia and tachyarrhythmias (19). The recommended dose is from 2 to 15 µg/kg/min and does not require renal adjustment (2).
Milrinone is a phosphodiesterase inhibitor that prevents the degradation of cyclic adenosine monophosphate (cAMP). In patients with heart failure, milrinone increases HR, SV and CO. It is also likely to decrease MAP, SVR and LVFP. It improves hemodynamics acutely; however, a concern exists regarding its long-term safety, as it has been correlated with new-onset atrial fibrillation and flutter and sustained hypotension. This is the rationale behind the use of milrinone only for patients with refractory cardiogenic shock (20). The recommended doses are 0.35–0.75 µg/kg/min and requires renal adjustment (2,20).
Levosimendan is a calcium-sensitizing agent, which exerts positive inotropic effects on the heart by increasing the cardiac contractile apparatus sensitivity to calcium. The SURVIVE randomized clinical trial that compared the efficacy between levosimendan and dobutamine in a total number of 1,327 patients revealed that levosimendan did not reduce the all-cause mortality compared to dobutamine alongside manifesting more peripheral vasodilating effects than dobutamine (21). Therefore, levosimendan’s role in cardiogenic shock is still not clear. The recommended dose is 0.05–0.2 µg/kg/min and should not be administered if SBP<90 mmHg (22).
Epinephrine increases SV and CO and decreases SVR by stimulating b2 receptors in the skeletal smooth muscle. It also increases pulmonary vascular resistance and right ventricular afterload. It is rarely used in acute decompensated heart failure because it increases the myocardial oxygen demand leading to ischemia (23).
Norepinephrine increases systemic BP, pulse pressure, peripheral vascular resistance and SV. In response to norepinephrine therapy, the CO is unchanged or decreased, and there is a compensatory vagal reflex that slows the HR. It is widely used as a first-line agent to increase BP and it is preferred rather than dopamine (24). The recommended starting dose is from 0.01 to 0.03mg/kg/min; maximum suggested dose is 0.1 mg/kg/min (2).
Dopamine is a catecholamine that is dose-dependent. At low doses, it causes vasodilation, especially in splanchnic and renal artery beds. At high doses, it promotes peripheral arterial and venous vasoconstriction. In patients with cardiogenic shock, dopamine increases HR, CO, SV and the left end-diastolic filling pressures whereas it reduces SVR (25). The SOAP II Investigators suggested that the patients with cardiogenic shock treated with dopamine had a higher 28-day mortality rate compared to those treated with norepinephrine (24). The ROSE Acute Decompensated Heart Failure randomized clinical trial has demonstrated that patients treated with low dose dopamine (2 mg/kg/min) had not reduced renal involvement or other clinical outcomes compared to them with nesiritide or placebo (26).
Hypovolemic (hemorrhagic) shock
One of the leading causes of mortality in the world is trauma. The main reason of death in these patients is uncontrolled hemorrhage (27). The first step in the management of the hemorrhage is to control bleeding along with fluid volume resuscitation in order to maintain adequate end-organ perfusion. However, an excessive amount of fluid administration can worsen hemorrhage. This vicious cycle is achieved due to the hemodilution of the coagulation factors leading to coagulopathy or due to hypothermia which precipitates coagulopathy as well (28). The use of vasopressors in hemorrhagic shock has been a controversial issue for a long period. Vasopressor administration could be a good option in hemorrhagic shock because it induces venous adrenergic stimulation which shifts blood from venous unstressed blood (“venous pooling”) to the circulation, thus maintaining adequate tissue perfusion. On the contrary, vasopressors induce arteriolar vasoconstriction with subsequent alterations of the microcirculation leading to tissue hypoxia (29). A retrospective study suggested that early vasopressor infusion in trauma patients increased the mortality rate independently of the trauma severity. The leading cause was low arterial pressure, increased fluid requirements and increased serum creatinine (30).
In the early stages of compensated hemorrhagic shock, the arterial pressure is maintained at adequate levels because of the compensatory sympathetic mechanisms that are activated, promoting venous and arterial vasoconstriction. When this mechanism becomes overwhelmed, the sympathetic system is not activated anymore and, subsequently, peripheral vascular resistance is reduced (31). Excessive NO production promotes vasodilation leading to vasoplegia (32). Even though vasoplegia describes excessive vasodilation, the term vascular hyporesponsiveness to vasopressors describes better the vascular state in shock (33). In this case, the use of vasopressors may be helpful.
Norepinephrine is the main first-line agent that is used due to its sympathomimetic properties. Inotropic support with dobutamine or epinephrine should be administered in the case of myocardial dysfunction which could be suspected if the patient does not respond in adequate fluid infusion and norepinephrine administration (34). It should be noted that vasopressors should be administered along with adequate fluids because norepinephrine alone could lead to detrimental effects (35).
Septic shock
Sepsis is a life-threatening dysfunction of the vital organs caused by the inability of the host to respond to an infection. Septic shock is a subset of sepsis accompanied by circulatory and cellular dysfunction that poses a life-threatening situation (36). Similarly to other emergencies, early sepsis diagnosis and management improves outcomes. Data from recent studies suggested that early initiation of vasopressors could prevent sustained hypotension leading to decreased mortality (37).
Fluid therapy is the first-line management of septic shock. Fluid resuscitation should be initiated promptly but with caution because positive fluid balance could lead to higher mortality rates (38). Thus, the use of norepinephrine in the first 2 hours of resuscitation could restrict the amount of fluid administration in the long-term management. Individualization of the management algorithms for septic shock is considered the most appropriate step by taking into account the diastolic arterial pressure of the patient; the lower the diastolic arterial pressure, the earlier the initiation of vasopressors (39).
As suggested by the sepsis surviving campaign (SSC), norepinephrine is the first vasopressor that should be administered in a case of septic shock (40). Norepinephrine and its venoconstricting effects move blood from the veins to the circulation increasing the preload which is of critical importance in early stages of septic shock as it can be overfilled during fluid administration (41). The MAP is the target for the resuscitation because it reflects the perfusion of the vital organs. A MAP >65mmHg is the resuscitation goal for the early stages of septic shock (40). Increasing MAP with norepinephrine leads to significant increase in the tissue oxygen saturation (StO2) recovery slope (a parameter that demonstrates the capacity of microvessels to be recruited in case of tissue hypoxia) (42). It has been postulated that an increase in MAP leads to increased peripheral microvascular blood flow. However, MAP is not always the most accurate parameter for the management of septic shock as often in sepsis there is dissociation between macrocirculation and microcirculation depending on the baseline characteristics of the patient (i.e., chronic hypertension) (43).
Norepinephrine is preferred rather than dopamine as recent studies have shown that the latter is responsible for tachyarrhythmias (44); it can be considered as an alternative solution only in highly selected patients with low risk of tachyarrhythmias or absolute bradycardia. A randomized control, double-blind clinical trial suggested that the use of epinephrine approximates the combined use of norepinephrine and dobutamine, but it demonstrated increased lactate levels in the peripheral tissues due to local ischemia (45). Thus, epinephrine is considered as an adjunct agent to norepinephrine if the MAP is not adequately increased.
Vasopressin is an endogenous peptide hormone released by the neurohypophysis promoting non-adrenergic vasoconstriction, especially in sepsis-associated hypotension. In the early phases of shock, the endogenous vasopressin stores become depleted. Vasopressin is used as a catecholamine-sparing agent to reduce the levels of norepinephrine dosage (46).
Inotropic support in septic shock patients is used when there is evidence of myocardial dysfunction as suggested by low CO, increased filling pressures and persistent hypoperfusion despite optimal fluid resuscitation and use of vasopressors. The myocardial dysfunction is common fact in sepsis; investigators suggest a possible self-protective, autoregulatory “hibernating” myocardium (47). Besides, the dysregulated cardiac contractility is a mixture of altered cellular metabolism and autoregulatory mechanisms in the microvasculature of the heart per se (48). Dobutamine is the first-line inotropic agent in septic patients as suggested by the SSC. Milrinone is recommended only in patients that are chronically beta-blocked or with chronic heart failure whose adrenergic receptors are desensitized (49). Levosimendan is not currently used in septic shock. Further investigations may shed light upon the use of agents that increase cytoplasmic calcium.
Anaphylactic shock
Anaphylaxis is a severe, life-threatening generalized or systemic hypersensitivity reaction that requires immediate and adequate care (50). In recent years, an increase in anaphylaxis incidence rates has occurred and, thus, more light should be shed upon this issue (51). Rapid recognition of anaphylaxis and prompt initiation of treatment is the cornerstone to reduce mortality rates. The first-line therapy is intramuscular injection of epinephrine, then removal of the allergen and, finally, monitoring of the airway, circulation, breathing and mental status (52).
Epinephrine is used as the first step of anaphylaxis treatment owing to its alpha-1 agonist effects that prevents airway edema, hypotension and, eventually, shock along with its beta-2 agonist effects that induces bronchodilation (6). However, as discussed above, epinephrine administration could have detrimental effects in patients with cardiovascular diseases. Moreover, endogenous and exogenous catecholamines play a significant role in the pathophysiology of Stress-Induced Cardiomyopathy (Takotsubo cardiomyopathy). Although rare, this mandates that epinephrine use should be monitored (especially since no guidelines suggest any other therapy as a substitute). The potential long-term adverse effects of epinephrine use in this kind of patients should be balanced with the acute management of the fatal risk of anaphylaxis (53). Besides, clinicians should keep in mind that anaphylaxis itself could affect the myocardium in a condition called the Kounis Syndrome (54). In any case, Guidelines recommend early epinephrine administration in every patient with anaphylaxis with no absolute contraindication in its use. The only considerations are in the elderly people that are beta-blocked due to the unopposed alpha-1 adrenergic effects and the reflex vagotonic effects.
Neurogenic shock
Neurogenic shock is a common cardiovascular dysfunction seen in the acute stage after spinal cord injury, characterized by significant hypotension as well as bradycardia. The changes in the hemodynamic profile are the result from the loss of supraspinal sympathetic excitatory input to sympathetic pre-ganglionic neurons, which are crucial for maintaining BP (55). The profound hypotension resulting from neurogenic shock leads to microvascular hypoperfusion of the spinal cord. This vicious cycle of microcirculation affects neurological recovery rates (56). Vasopressor therapy depends on the level of the injury and the patient’s hemodynamic profile.
According to the Consortium for Spinal Cord Medicine, injuries at the cervical level and at the thoracic until T6 demand a vasopressor agent with both inotropic and vasoconstrictive properties to support both the vessel tone and cardiac contractility given the fact that sympathetic innervation of the heart originates at the level T1–T4 (57). Thus, dopamine and norepinephrine are the agents used at this specific injury level. Injuries in lower thoracic and lumbar level warrant the need of a specific peripheral vasoconstrictor such as phenylephrine, which acts only on alpha-1 receptors. Epinephrine is not widely used in neurogenic shock because it exerts its beta-1 adrenergic effects leading to arrhythmias requiring continuous monitoring (58). Dobutamine is not used in neurogenic shock because of its peripheral vasodilatory properties and potential reflex bradycardia (59).
The most common inotropic and vasopressor agents used in everyday clinical practice for shocked patients, their receptors, actions and role in therapy are presented in Table 1.
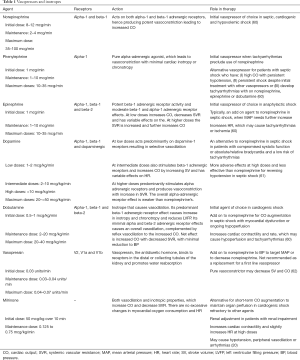
Full table
Thoughts for the future
Fluid resuscitation, vasopressors and inotropes are the first line medication for the different types of shock due to their mechanism of action and their well-established clinical outcome (2,9). However, as experimental research and clinical trials are constantly being published, new suggestions appear regarding alterations and novelties in the use of these medications in the management of shock (Tables 2,3).
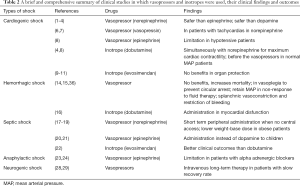
Full table

Full table
Clinical advances
Regarding cardiogenic shock recent studies suggest that it is more helpful to use norepinephrine instead of epinephrine in patient with cardiogenic shock due to epinephrine’s many harmful effects such us elevation of organ damage biomarkers and lactate levels or even increasing of mortality probability. For these reasons, there is a trend to use norepinephrine as a safer agent in the case of cardiogenic shock (1,2). One more comparison between norepinephrine and dopamine revealed that norepinephrine remains the most appropriate medication and first line drug in cardiogenic shock due to the fact that the dopamine produces more arrhythmic events in patients and carries a higher mortality rate (3,4). Recent data suggest norepinephrine as a more useful drug in cardiogenic shock instead of the broadly used epinephrine and dopamine. Regarding vasopressin, no new recommendations can be made as there are no clinical trials comparing it to norepinephrine (1,4,5). However, there are some indications suggesting treating patients with vasopressin instead of norepinephrine when the second one produces detrimental tachycardias (6,7). Furthermore, there is a limitation in epinephrine administration in hypotensive patients (8).
As far as inotropes are concerned, it is well established that dobutamine is administrated at the same time with norepinephrine in order to maximize the contraction of the heart. On the other hand, no novel data regarding inotropes, such as levosimendan and phosphodiesterase inhibitors, have been published, suggesting no new perspectives in cardiogenic shock, except for some new evidence that failed to report any benefit from levosimendan in organ protection and mortality rate (4,8-11). The inotropic support in patients with hypotension or vasoplegia must be started after vasopressors such us norepinephrine; however, in case of patients with normal BP, the first step is the use of inotropes (dobutamine for cardiogenic shock) (6). So, vasopressors and inotropes are the initial treatment, but the strategy depends on the clinical status of patients (12).
Regarding vasopressors in hemorrhagic shock, there is a lot of debate about their use due to the increase in mortality rate after early administration (13). However, in cases of vasoplegia, the use of vasopressors is mandatory to prevent circulatory arrest. The most recent recommendations advocate the administration of vasopressors to keep the BP in the desirable range when there is no response to fluid resuscitation (14,15). Furthermore, in hemorrhage, a small dose of vasopressin is capable to maintain BP, when fluid therapy is not adequate. When myocardial dysfunction is present, inotropes such as dobutamine are administrated (16). Finally, despite the disadvantages that may appear from the use of vasopressors in hemorrhagic shock, there is splanchnic vasoconstriction, hence reducing bleeding by lowering portal output (14).
Vasopressors and inotropes as well are still the first-choice medication for septic shock. Norepinephrine is the main treatment choice as it is mentioned before. The central administration of norepinephrine is the main strategy but in case of patients that there is no immediate central access, it is possible to insert vasopressors peripherally but for a short period of time (17). Furthermore, in case of obese patients the treatment with norepinephrine can be done with lower weight-base dose (18,19). For septic shock in children, epinephrine is suggested recently as a first choice instead of dopamine (20,21). Finally, there is one more suggestion regarding inotropes that shows the ability of levosimendan to produce better clinical outcomes in comparison with dobutamine, however without decreasing mortality (22).
In anaphylactic shock, the general principles of management remain the same, which is the administration of epinephrine as a first line medication, with some noticeable exceptions of resistance to epinephrine in patients with alpha adrenergic blockers (23-25). For neurogenic shock, as mentioned previously, except for fluid resuscitation, the treatment is administration of vasopressors like epinephrine, norepinephrine and dopamine. In cases with slower recovery, the intravenous use of vasopressors should be continued for longer periods (26-30).
Experimental advances
In cardiogenic shock research, data from different animal models such as dogs, pigs and rats indicated that the use of vasopressors was harmful increasing mortality rate (31). Furthermore, in case of cardiogenic shock due to poisoning, a porcine model demonstrated that adding norepinephrine to the standard therapeutic scheme maximized the treatment effect (32). Most recently, porcine hemorrhagic shock models revealed that the combined use of norepinephrine and vasopressin produced beneficial effects restoring and maintaining the arterial pressure independently of volume resuscitation; however, lactic acidosis was not prevented (33). A rat model indicated the beneficial effects of vasopressors when administering norepinephrine in hemorrhagic shock, following the restriction of fluid therapy, as it improved end-organ perfusion (34). Finally, a recent meta-analysis regarding hemorrhagic shock animal studies indicated that vasopressin was more efficient than the rest of the vasopressor agents and, thus, it could be an alternative option as a first step in hemorrhagic shock treatment (35).
New agents
To complicate things even further, novel inotropic agents are being constantly developed and tested (sometimes with not encouraging results) in the experimental and clinical setting with hopes of implementing them in future treatment Guidelines.
Istaroxime is a Na+/K+ ATPase inhibitor and activator of the sarcoplasmic reticulum calcium pump that showed positive inotropic and lusitropic effect, but with no vasodilatory properties. Omecamtivmecarbil, a cardiac myosin activator, increased myocardial contractile force; improvements in cardiac function were not associated with increased myocardial oxygen consumption when tested on canine models. Nitroxyl, a molecule very similar to NO, showed, in animal and in vitro studies, potent arterio- and veno-dilating effects without tolerance or tachyphylaxis, as well as positive inotropic and lusitropic effects. Lastly, gene therapy focusing on increasing sarcoplasmic reticulum calcium pump activity has shown improvement in systolic and diastolic functions in different animal models of heart failure (63-66).
Conclusions
Shock, a profound hemodynamic impairment in which oxygen delivery to the tissues are not sufficient to meet metabolic requirements, is characterized by inadequate tissue perfusion, resulting in life-threatening impairment of oxygen and nutrient delivery Treatment of shock consists of identifying and reversing the underlying pathogenesis and correcting hemodynamic abnormalities. Vasopressors should be initiated in refractory hypotension despite adequate fluid and/or blood product resuscitation following the SSC guidelines recognizing that a MAP of 60 to 65 mmHg is required to promote adequate perfusion pressure for organs. If after appropriate fluid resuscitation, MAP does not improve to about 60 mmHg, it is recommended to administer vasopressors (40). In low CO states, the use of an inotropic agent should be considered. Norepinephrine continues to be the first-line agent for blood pressure support. Vasopressin should be considered to decrease the dose of norepinephrine or augment the MAP with a goal of ≥65 mmHg. Epinephrine is considered the second-line agent. Dopamine should be considered instead of norepinephrine only in patients with relative or absolute bradycardia who have a low risk of tachyarrhythmias. Dobutamine is still recommended for patients with persistent hypoperfusion despite adequate intravascular volume and vasopressor administration.
Even though the pharmacokinetic and pharmacodynamics properties of inotropes and vasopressors are well described, in clinical practice, their net effect is not always as anticipated by the treating physician. During critical illness, alterations in receptor and intracellular signaling pathways, formation of free radicals and the auto-oxidation of catecholamines, alterations in regional blood flow, metabolic changes, bacterial growth, immune system alterations mediated by apoptosis, inflammation and other receptor systems, alterations in the coagulation cascade and platelets alongside the altered mobility of the gastrointestinal tract can present with patients not responding to the agents administered or, sometimes, having opposite or undesired effects (67).
We deliberately chose not to include in this review specific indications in critically ill patients that require administration of specific agents (i.e., metaraminol, a mixed direct- and indirect-acting sympathomimetic that has direct action at alpha-adrenoceptors and increases the amount of noradrenaline in the synaptic cleft mainly used during surgery or critical illness to reverse short-term episodes of hypotension, dopexamine, the most frequently investigated agent in the surgical population, which even though may have specific beneficial effects on tissue microvascular flow and oxygenation in patients following major gastrointestinal surgery, its b2-agonist effects may result in significant tachycardia at higher doses and increased incidence of myocardial ischemia, isoproterenol, a non-selective β-adrenergic agonist reducing systemic and pulmonary vascular resistance causing a fall in mean arterial and diastolic BP while systolic pressure remaining unchanged or rising modestly due to increased CO, but with limited clinical use, except for patients after heart transplantation, because of significant adverse effects such as tachycardia and arrhythmias, or enoximone, an imidazolone and a selective phosphodiesterase III which while in vivo demonstrates less inotropy and chronotropy, but more lusitropy when compared with milrinone, at clinical doses, does not produce significant inotropic and lusitropic effects) (40).
The Critical Care Practice Committee of the Canadian Association of Emergency Physician in an attempt to guide the emergency medicine physician with vasopressor or inotrope use in shock states in the emergency setting published Evidence Based Consensus Guidelines, where apart from offering clear indications for vasopressor or inotrope use, gives information on how the aforementioned agents should be administered to the ED patients (68).
Despite widespread use, the evidence base for the use of inotropes and vasopressors in critically ill and shocked patients is limited. This is due to the few large randomized controlled trials directly comparing agents in terms of patient survival. Clearly, many patients would not survive without inotropic/vasopressor support, but there is, nonetheless, considerable variation in clinical practice. Current practice can be improved through a more detailed understanding of the diverse actions of these agents and the potential toxic effects. It would seem prudent to use minimum necessary doses of such agents until our evidence base improves.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Cecconi M, Hernandez G, Dunser M, et al. Fluid administration for acute circulatory dysfunction using basic monitoring: narrative review and expert panel recommendations from an ESICM task force. Intensive Care Med 2019;45:136. [Crossref] [PubMed]
- Hollenberg SM. Vasoactive drugs in circulatory shock. Am J Respir Crit Care Med 2011;183:847-55. [Crossref] [PubMed]
- Spahn DR, Bouillon B, Cerny V, et al. Management of bleeding and coagulopathy following major trauma: an updated European guideline. Crit Care 2019;23:98. [Crossref] [PubMed]
- Hochman JS, Sleeper LA, Webb JG, et al. Early revascularization and long-term survival in cardiogenic shock complicating acute myocardial infarction. JAMA 2006;295:2511-5. [Crossref] [PubMed]
- Sharawy N, Lehmann C. New directions for sepsis and septic shock research. J Surg Res 2015;194:520-7. [Crossref] [PubMed]
- Lee SE. Management of Anaphylaxis. Otolaryngol Clin North Am 2017;50:1175-84. [Crossref] [PubMed]
- Hadley MN, Walters BC, Grabb PA, et al. Guidelines for the management of acute cervical spine and spinal cord injuries. Clin Neurosurg 2002;49:407-98. [PubMed]
- Torsvik M, Gustad LT, Mehl A, et al. Early identification of sepsis in hospital inpatients by ward nurses increases 30-day survival. Crit Care 2016;20:244. [Crossref] [PubMed]
- Vincent JL, Weil MH. Fluid challenge revisited. Crit Care Med 2006;34:1333-7. [Crossref] [PubMed]
- Gamper G, Havel C, Arrich J, et al. Vasopressors for hypotensive shock. Cochrane Database Syst Rev 2016;2:CD003709. [PubMed]
- Ahlquist RP. A study of the adrenotropic receptors. Am J Physiol 1948;153:586-600. [Crossref] [PubMed]
- Allwood MJ, Cobbold AF, Ginsburg J. Peripheral vascular effects of noradrenaline, isopropylnoradrenaline and dopamine. Br Med Bull 1963;19:132-6. [Crossref] [PubMed]
- Williamson AP, Seifen E, Lindemann JP, et al. WB4101- and CEC-sensitive positive inotropic actions of phenylephrine in rat cardiac muscle. Am J Physiol 1994;266:H2462-7. [PubMed]
- Reynolds HR, Hochman JS. Cardiogenic shock current concepts and improving outcomes. Circulation 2008;117:686-97. [Crossref] [PubMed]
- Pöss J, Desch S, Thiele H. Shock management in acute myocardial infarction. EuroIntervention 2014;10:T74-82. [Crossref] [PubMed]
- Basir MB, Kapur NK, Patel K, et al. Improved Outcomes Associated with the use of Shock Protocols: Updates from the National Cardiogenic Shock Initiative. Catheter Cardiovasc Interv 2019;93:1173-83. [PubMed]
- Mebazaa A, Combes A, van Diepen S, et al. Management of cardiogenic shock complicating myocardial infarction. Intensive Care Med 2018;44:760-73. [Crossref] [PubMed]
- Amado J, Gago P, Santos W, et al. Cardiogenic shock: Inotropes and vasopressors. Rev Port Cardiol 2016;35:681-95. [Crossref] [PubMed]
- Abraham WT, Adams KF, Fonarow GC, et al. In-hospital mortality in patients with acute decompensated heart failure requiring intravenous vasoactive medications: an analysis from the Acute Decompensated Heart Failure National Registry (ADHERE). J Am Coll Cardiol 2005;46:57-64. [Crossref] [PubMed]
- Cuffe MS, Califf RM, Adams KF Jr, et al. Short-term intravenous milrinone for acute exacerbation of chronic heart failure: a randomized controlled trial. JAMA 2002;287:1541-7. [Crossref] [PubMed]
- Mebazaa A, Nieminen MS, Packer M, et al. Levosimendan vs dobutamine for patients with acute decompensated heart failure: the SURVIVE Randomized Trial. JAMA 2007;297:1883-91. [Crossref] [PubMed]
- Follath F, Franco F, Cardoso JS. European experience on the practical use of levosimendan in patients with acute heart failure syndromes. Am J Cardiol 2005;96:80G-5G. [Crossref] [PubMed]
- Møller MH, Claudius C, Junttila E, et al. Scandinavian SSAI clinical practice guideline on choice of first-line vasopressor for patients with acute circulatory failure. Acta Anaesthesiol Scand 2016;60:1347-66. [Crossref] [PubMed]
- De Backer D, Biston P, Devriendt J, et al. Comparison of dopamine and norepinephrine in the treatment of shock. N Engl J Med 2010;362:779-89. [Crossref] [PubMed]
- Nativi-Nicolau J, Selzman CH, Fang JC, et al. Pharmacologic therapies for acute cardiogenic shock. Curr Opin Cardiol 2014;29:250-7. [Crossref] [PubMed]
- Chen HH, Anstrom KJ, Givertz MM, et al. Low-dose dopamine or low-dose nesiritide in acute heart failure with renal dysfunction: the ROSE acute heart failure randomized trial. JAMA 2013;310:2533-43. [Crossref] [PubMed]
- Cothren CC, Moore EE, Hedegaard HB, et al. Epidemiology of urban trauma deaths: A comprehensive reassessment 10 years later. World J Surg 2007;31:1507-11. [Crossref] [PubMed]
- Gupta B, Garg N, Ramachandran R. Vasopressors: Do they have any role in hemorrhagic shock? J Anaesthesiol Clin Pharmacol 2017;33:3-8. [Crossref] [PubMed]
- Harrois A, Baudry N, Huet O, et al. Norepinephrine Decreases Fluid Requirements and Blood Loss While Preserving Intestinal Villi Microcirculation during Fluid Resuscitation of Uncontrolled Hemorrhagic Shock in Mice. Anesthesiology 2015;122:1093-102. [Crossref] [PubMed]
- Plurad DS, Talving P, Lam L, et al. Early vasopressor use in critical injury is associated with mortality independent from volume status. J Trauma 2011;71:565-70; discussion 570-2. [Crossref] [PubMed]
- Beloncle F, Meziani F, Lerolle N, et al. Does vasopressor therapy have an indication in hemorrhagic shock? Ann Intensive Care 2013;3:13. [Crossref] [PubMed]
- Thiemermann C, Szabó C, Mitchell JA, et al. Vascular hyporeactivity to vasoconstrictor agents and hemodynamic decompensation in hemorrhagic shock is mediated by nitric oxide. Proc Natl Acad Sci U S A 1993;90:267-71. [Crossref] [PubMed]
- Levy B, Fritz C, Tahon E, et al. Vasoplegia treatments: the past, the present, and the future. Crit Care 2018;22:52. [Crossref] [PubMed]
- Hamada SR, Gauss T, Pann J, et al. European trauma guideline compliance assessment: The ETRAUSS study. Crit Care 2015;19:423. [Crossref] [PubMed]
- Tsuneyoshi I, Onomoto M, Yonetani A, et al. Low-dose vasopressin infusion in patients with severe vasodilatory hypotension after prolonged hemorrhage during general anesthesia. J Anesth 2005;19:170-3. [Crossref] [PubMed]
- Angus DC, Linde-Zwirble WT, Lidicker J, et al. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 2001;29:1303-10. [Crossref] [PubMed]
- Bai X, Yu W, Ji W, et al. Early versus delayed administration of norepinephrine in patients with septic shock. Crit Care 2014;18:532. [Crossref] [PubMed]
- Marik PE, Linde-Zwirble WT, Bittner EA, et al. Fluid administration in severe sepsis and septic shock, patterns and outcomes: an analysis of a large national database. Intensive Care Med 2017;43:625-32. [Crossref] [PubMed]
- Hamzaoui O, Scheeren TWL, Teboul J. Norepinephrine in septic shock. Curr Opin Crit Care 2017;23:342-7. [Crossref] [PubMed]
- Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med 2017;43:304-77. [Crossref] [PubMed]
- Persichini R, Silva S, Teboul JL, et al. Effects of norepinephrine on mean systemic pressure and venous return in human septic shock. Crit Care Med 2012;40:3146-53. [Crossref] [PubMed]
- Georger JF, Hamzaoui O, Chaari A, et al. Restoring arterial pressure with norepinephrine improves muscle tissue oxygenation assessed by near-infrared spectroscopy in severely hypotensive septic patients. Intensive Care Med 2010;36:1882-9. [Crossref] [PubMed]
- Hernández G, Teboul JL. Is the macrocirculation really dissociated from the microcirculation in septic shock? Intensive Care Med 2016;42:1621-24. [Crossref] [PubMed]
- Rui Q, Jiang Y, Chen M, et al. Dopamine versus norepinephrine in the treatment of cardiogenic shock: A PRISMA-compliant meta-analysis. Medicine (Baltimore) 2017;96:e8402. [Crossref] [PubMed]
- Annane D, Vignon P, Renault A, et al. Norepinephrine plus dobutamine versus epinephrine alone for management of septic shock: a randomised trial. Lancet 2007;370:676-84. [Crossref] [PubMed]
- Stratton L, Berlin DA, Arbo JE. Vasopressors and Inotropes in Sepsis. Emerg Med Clin North Am 2017;35:75-91. [Crossref] [PubMed]
- Levy RJ, Piel DA, Acton PD, et al. Evidence of myocardial hibernation in the septic heart. Crit Care Med 2005;33:2752-6. [Crossref] [PubMed]
- Hunter JD, Doddi M. Sepsis and the heart. Br J Anaesth 2010;104:3-11. [Crossref] [PubMed]
- Overgaard CB, Dzavík V. Inotropes and vasopressors: review of physiology and clinical use in cardiovascular disease. Circulation 2008;118:1047-56. [Crossref] [PubMed]
- Simons FE, Ardusso LR, Dimov V, et al. World Allergy Organization Anaphylaxis Guidelines: 2013 update of the evidence base. Int Arch Allergy Immunol 2013;162:193-204. [Crossref] [PubMed]
- Drupad HS, Nagabushan H. Level of knowledge about anaphylaxis and its management among health care providers. Indian J Crit Care Med 2015;19:412-5. [Crossref] [PubMed]
- Wood RA, Camargo CA, Lieberman P, et al. Anaphylaxis in America: the prevalence and characteristics of anaphylaxis in the United States. J Allergy Clin Immunol 2014;133:461-7. [Crossref] [PubMed]
- Lieberman P, Simons FER. Anaphylaxis and cardiovascular disease: therapeutic dilemmas. Clin Exp Allergy 2015;45:1288-95. [Crossref] [PubMed]
- Kounis NG. Coronary hypersensitivity disorder: the kounis syndrome. Clin Ther 2013;35:563-71. [Crossref] [PubMed]
- Krassioukov A, Claydon VE. The clinical problems in cardiovascular control following spinal cord injury: An overview. Prog Brain Res 2006;152:223-9. [Crossref] [PubMed]
- Furlan JC, Fehlings MG, Shannon P, et al. Descending vasomotor pathways in humans: correlation between axonal preservation and cardiovascular dysfunction after spinal cord injury. J Neurotrauma 2003;20:1351-63. [Crossref] [PubMed]
- Wing PC. Early acute management in adults with spinal cord injury: a clinical practice guideline for health-care providers. Who should read it? J Spinal Cord Med. 2008;31:360. [Crossref] [PubMed]
- Hadley MN, Walters BC, Grabb PA, et al. Blood pressure management after acute spinal cord injury. Neurosurgery 2002;50:S58-62. [Crossref] [PubMed]
- Ball PA. Critical Care of Spinal Cord Injury. Spine (Phila Pa 1976) 2001;26:S27-30. [Crossref] [PubMed]
- Hollenberg SM. Inotrope and vasopressor therapy of septic shock. Crit Care Clin 2009;25:781-802. ix. [Crossref] [PubMed]
- Al-Hesayen A, Azevedo ER, Newton GE, et al. The effects of dobutamine on cardiac sympathetic activity in patients with congestive heart failure. J Am Coll Cardiol 2002;39:1269-74. [Crossref] [PubMed]
- Mutlu G, Factor P. Role of vasopressin in the management of septic shock. Intensive Care Med 2004;30:1276-91. [Crossref] [PubMed]
- Francis GS, Bartos JA, Adatya S. Inotropes. J Am Coll Cardiol 2014;63:2069-78. [Crossref] [PubMed]
- Hasenfuss G, Teerlink JR. Cardiac inotropes: current agents and future directions. Eur Heart J 2011;32:1838-45. [Crossref] [PubMed]
- Garg V, Frishman WH. A new approach to inotropic therapy in the treatment of heart failure: cardiac myosin activators in treatment of HF. Cardiol Rev 2013;21:155-9. [Crossref] [PubMed]
- Nagy L, Pollesello P, Papp Z. Inotropes and inodilators for acute heart failure: sarcomere active drugs in focus. J Cardiovasc Pharmacol 2014;64:199-208. [Crossref] [PubMed]
- Bangash MN, Kong ML, Pearse RM. Use of inotropes and vasopressor agents in critically ill patients. Br J Pharmacol 2012;165:2015-33. [Crossref] [PubMed]
- Djogovic D, MacDonald S, Wensel A, et al. Vasopressor and inotrope use in Canadian emergency departments: evidence based consensus guidelines. CJEM 2015;17 Suppl 1:1-16. [Crossref]
Cite this article as: Manolopoulos PP, Boutsikos I, Boutsikos P, Iacovidou N, Ekmektzoglou K. Current use and advances in vasopressors and inotropes support in shock. J Emerg Crit Care Med 2020;4:20.


