
Acquired hemophilia A presenting as spontaneous hemorrhage following corpus luteum cyst rupture: a case report and review of the literature
Introduction
Acquired hemophilia A (AHA) is a rare and life-threatening hematologic disorder by which autoantibodies target and inhibit circulating factor VIII (1). The incidence of this condition is only about 1 to 4 people per million per year, with a mortality rate estimated to be as high as 22% (2). In contrast with hereditary hemophilia, which is X-linked and therefore almost exclusively affects males, acquired hemophilia does not predominate in one sex over the other (3). Approximately 50% of AHA cases can be attributed to secondary causes such as autoimmune diseases, cancers, drug reactions, or pregnancy, with the remaining 50% being isolated or indeterminate (4). AHA is most common among the elderly, with a median age between 64 to 78 years old. Cases that arise in younger demographics are more commonly associated with pregnancy and autoimmune diseases (1).
The initial presentation of AHA can be vast, spanning from spontaneous and persistent bleeding to post-operative or traumatic-related hemorrhage (3). In cases of bleeding in tight spaces, such as deep to the muscle fascia, compartment syndrome can arise (5,6), while bleeding within the brain can result in intracranial hemorrhage (7-13). Interestingly, although bleeding may be the predominant concern in many of these instances, mortality as a direct result of bleeding is less common (17.2% of all deaths), possibly due to timely and continuous volume resuscitation interventions (14). In cases when bleeding is not a direct cause of death, however, it often leads to significant morbidity and prolonged hospitalization (1). Given this, appropriate medical treatment is imperative.
As with all causes of uncontrolled bleeding, appropriate hemostasis is essential. However, with regards to AHA, not only is controlling the bleeding vital; but so is identifying the condition so that immunosuppressive treatment to eradicate the inhibitor can be initiated as soon as possible (15).
We present a case of AHA in a patient who presented to the emergency department (ED) with a ruptured corpus luteum cyst. Given the rarity of this condition and the necessity for rapid diagnosis and appropriate management, we discuss the diagnostic workup of AHA to guide the emergency medicine and inpatient team in treating patients with this rare condition.
We present the following article in accordance with the CARE reporting checklist (available at http://dx.doi.org/10.21037/jeccm-20-144).
Case presentation
A 25-year-old female with no significant past medical history presented to the ED complaining of lower abdominal pain following a syncopal event. The patient was tachycardic and hypotensive, with physical exam demonstrating ecchymosis of the upper extremities bilaterally. Laboratory results revealed leukocytosis and anemia, with hemoglobin of 6.2 g/dL. β-human chorionic gonadotropin level (βHCG) was below 5 mIU/mL. The patient noted fatigue and weakness, but denied fever, chills, nausea, vomiting, melena, blood in the stool, or any recent history of trauma. She reported no surgical history, no prior history of easy bruising or bleeding, and no family history of bleeding disorders. She is nulliparous with her last menstrual period being three and a half weeks prior.
A computed tomography (CT) scan of the abdomen/pelvis demonstrated hemoperitoneum with extravasation in the deep pelvis, likely related to gynecologic organs. Obstetrics/Gynecology was consulted and performed an emergent laparoscopy, 1,500 mL of hemoperitoneum was evacuated. The left ovary contained a hemorrhagic corpus luteum cyst, which was removed. No active source of bleeding was found.
Coagulation studies revealed a prolonged activated partial thromboplastin time (aPTT) of 97 seconds. Hematology was consulted and the patient was started on recombinant Factor VIIa (FVIIa), fresh frozen plasma (FFP), cryoprecipitate, desmopressin, and prothrombin complex concentrate (PCC) to circumvent any clotting cascade abnormality. The patient, however, became hemodynamically unstable again and an emergent exploratory laparotomy was performed. A large left abdominal wall hematoma was found; however no other source of bleeding was identified. Further laboratory studies including aPTT mixing study showed a failure to correct, suggesting the presence of a coagulation inhibitor. Factor VIII (FVIII) activity level was extremely low at <1%, and FVIII Inhibitor level was exceedingly high at 141 Bethesda units, confirming the diagnosis of AHA.
The patient was started on immunosuppressive therapy consisting of high dose glucocorticoids, intravenous immunoglobulin G, Cyclophosphamide once weekly x 6 doses, and Rituximab and Bortezomib once weekly x 3 doses. Removal of the FVIII Inhibitor was also attempted with plasma exchange daily. Repeat CT of the Abdomen/Pelvis on day 9 revealed multiple large subcutaneous hematomas throughout the anterior abdominal wall along the abdominal wound incision and a large intramuscular hematoma involving the entire left abdominal rectus muscle; evacuation of the hemoperitoneum was also noted (Figure 1).
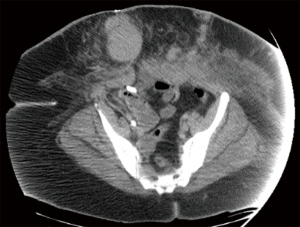
Approximately two weeks into the hospital course, the patient demonstrated clinical improvement. She was extubated on day 13 and FVIIa was discontinued. On day 17, FVIII Inhibitor level decreased to 25 from 141. The patient’s hospital course was further complicated by abdominal wound infection and bacteremia, with cultures demonstrating Proteus mirabilis and Serratia marcescens, respectively. During this course, the patient was also diagnosed with mixed connective tissue disease (MCTD). The patient was hospitalized for 43 days, during which she received a total of 41 units of PRBCs, 3 units of platelets, and was exchanged 196 of units of FFP over 34 days (Figure 2). Upon discharge, her FVIII level had increased to 22% from less than 1%. Following discharge, the patient has continued rituximab, cyclophosphamide and prednisone x 3 cycles. Furthermore, her factor VII inhibitor levels remain undetectable, while her Factor VIII level is up to 45%. At the time of publication, the patient remains in outpatient follow-up with no residual sequelae.

All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Discussion
Hemorrhagic shock is a relatively common pathology encountered in the ED. Most commonly, these situations arise from hemorrhage in a patient with normal hemostatic capabilities. Occasionally, bleeding disorders may be discovered in patients who describe a personal or family history of bleeding. However, in some instances, as in this case, a patient can acquire a bleeding disorder and present with a single life-threatening bleeding episode.
We present such an instance in a female of reproductive age who developed AHA and presented with profound bleeding following a ruptured corpus luteum cyst. Gynecologic emergencies are common in the ED, with ruptured corpus luteum cysts and ectopic pregnancies being the most common causes of spontaneous gynecologic hemoperitoneum (16). Although spontaneous bleeding following ovulation is relatively benign and without significant clinical consequences in otherwise healthy women, sustained hemorrhage, especially in those with bleeding disorders, can result in life threatening hemoperitoneum (17,18).
In AHA, the only initial diagnostic clue may be an isolated elevation of aPTT. Upon discovery of any coagulation study abnormality in a patient with severe bleeding with circulatory compromise, a hematologist should be immediately consulted. An aPTT mixing study should be ordered next; correction of the aPTT should raise suspicion of an intrinsic clotting factor deficiency, while persistently elevated aPTT (as in our patient) indicates either an acquired coagulation inhibitor or lupus anticoagulant related etiology. Final laboratory testing involves measuring clotting factor inhibitor levels and testing for lupus anticoagulant (19).
The first step in managing these severe bleeding episodes in a case of suspected AHA is administering circumferential clotting cascade agents, including activated prothrombin complex concentrates (APCCs), human FVIII and recombinant activated factor VIIa (19). In addition, as the etiology of this disorder is autoimmune, it is imperative to promptly initiate immunosuppressive therapy to suppress the body’s production of the clotting factor inhibitor. Immunosuppressive therapy may include high dose glucocorticoids, intravenous immunoglobulin G, and various chemotherapeutic drugs (1). Lastly, plasmapheresis may be utilized in severe cases such as this one.
When treating patients with AHA, careful attention must be made to any signs of infection or sepsis, as infectious diseases account for nearly half of AHA related mortalities (20). This is especially important if the patient has previously diagnosed AHA and is on any immunosuppressive treatment regimen (21). In addition, given patients with AHA lack normal hemostatic capabilities, the risks and benefits for all invasive procedures must be carefully considered. In the case of our patient, the majority of her bleeding sequelae was a result of bleeding into her abdominal wall surgical incisions. Hematoma accumulation at the site of her internal jugular central line and mild oozing from her peripheral intravenous lines also contributed to her deteriorating condition. With regards to the patient’s hemodynamic compromise and indeterminate source of bleeding, emergent surgical exploration was indicated for this patient. However, we recommend physicians complete thorough hematologic workup in patients who present with acute uncontrolled bleeding of an unknown primary source in the absence of traumatic injury or history of hematologic disorders, as even minimally invasive procedures in patients with AHA can be catastrophic.
Conclusions
Swift recognition and diagnosis of the underlying bleeding disorder is essential to achieving favorable outcomes in patients with acquired hemophilia. This is only possible through an appropriate history and physical examination and ordering the appropriate hematologic studies. Delays in any of these steps will not only prolong the time to effective treatment, but can be the determining factor in patient outcome, even after hospital admission. The emergency physician is therefore instrumental in dictating outcomes in patients with acquired hemophilia who present with spontaneous bleeding without an identifiable cause. Acute treatment of this condition should not be delayed, and should focus on volume resuscitation and rapid hemostasis, with the eventual goal to achieve immunosuppression in order to eliminate the coagulation factor inhibitor.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at http://dx.doi.org/10.21037/jeccm-20-144
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm-20-144). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kruse-Jarres R, Kempton CL, Baudo F, et al. Acquired hemophilia A: Updated review of evidence and treatment guidance. Am J Hematol 2017;92:695-705. [Crossref] [PubMed]
- Franchini M, Lippi G. Acquired factor VIII inhibitors. Blood 2008;112:250-5. Erratum in: Blood 2009;113:5368. [Crossref] [PubMed]
- Baudo F, Caimi T, de Cataldo F. Diagnosis and treatment of acquired haemophilia. Haemophilia 2010;16:102-6. [Crossref] [PubMed]
- Baudo F, de Cataldo F. The problem of acquired hemophilia. Blood 2015;125:1052-3. [Crossref] [PubMed]
- Van Laer M, Penaloza A, Stockman W, et al. Compartment syndrome of the forearm with life-threatening bleeding after fasciotomy as the presenting sign of postpartum acquired hemophilia A: a case report. Blood Coagul Fibrinolysis 2019;30:120-6. [Crossref] [PubMed]
- Alidoost M, Conte GA, Chaudry R, et al. A Unique Presentation of Spontaneous Compartment Syndrome due to Acquired Hemophilia A and Associated Malignancy: Case Report and Literature Review. World J Oncol 2020;11:72-5. [Crossref] [PubMed]
- Bonnaud I, Saudeau D, de Toffol B, et al. Recurrence of spontaneous subdural haematoma revealing acquired haemophilia. Eur Neurol 2003;49:253-4. [Crossref] [PubMed]
- Akamatsu Y, Hayashi T, Yamamoto J, et al. Newly Diagnosed Acquired Hemophilia A Manifesting as Massive Intracranial Hemorrhage Following a Neurosurgical Procedure. World Neurosurg 2018;111:175-80. [Crossref] [PubMed]
- Micic D, Williams EC, Medow JE. Cerebellar hemorrhage as a first presentation of acquired Hemophilia A. Neurocrit Care 2011;15:170-4. [Crossref] [PubMed]
- Tsuyama N, Ichiba T, Naito H. Unusual Initial Manifestation of Acquired Hemophilia A: A Normal Activated Partial Thromboplastin Time, Intramuscular Hematoma and Cerebral Hemorrhage. Intern Med 2016;55:3347-9. [Crossref] [PubMed]
- Marquardt L, Haubelt H, Gass S, et al. Intracranial bleeding in acquired hemophilia. Nervenarzt 2006;77:1480-2. [Crossref] [PubMed]
- Mamoli B, Sonneck G, Lechner K. Intracranial and spinal hemorrhage in haemophilia (author’s transl). J Neurol. 1976;211:143-54. [Crossref] [PubMed]
- Saito R, Takahashi T, Endo H, et al. A case of subarachnoid hemorrhage complicated by acquired hemophilia. No Shinkei Geka 2009;37:1215-9. [PubMed]
- Knoebl P, Marco P, Baudo F, et al. Demographic and clinical data in acquired hemophilia A: results from the European Acquired Haemophilia Registry (EACH2). J Thromb Haemost 2012;10:622-31. [Crossref] [PubMed]
- Zeitler H, Ulrich-Merzenich G, Goldmann G, et al. The relevance of the bleeding severity in the treatment of acquired haemophilia - an update of a single-centre experience with 67 patients. Haemophilia 2010;16:95-101. [Crossref] [PubMed]
- Lubner M, Menias C, Rucker C, et al. Blood in the belly: CT findings of hemoperitoneum. Radiographics 2007;27:109-25. [Crossref] [PubMed]
- Gupta N, Dadhwal V, Deka D, et al. Corpus luteum hemorrhage: rare complication of congenital and acquired coagulation abnormalities. J Obstet Gynaecol Res 2007;33:376-80. [Crossref] [PubMed]
- Hoffman R, Brenner B. Corpus luteum hemorrhage in women with bleeding disorders. Womens Health (Lond) 2009;5:91-5. [Crossref] [PubMed]
- Xu L, Chen J, Zhou X, et al. Acquired hemophilia A presenting as progressive intra-abdominal hemorrhage, muscle hemorrhage and hemothorax postpartum: A case report and literature review. Exp Ther Med 2019;17:633-8. [PubMed]
- Federici AB, Budde U, Castaman G, et al. Current diagnostic and therapeutic approaches to patients with acquired von Willebrand syndrome: a 2013 update. Semin Thromb Hemost 2013;39:191-201. [Crossref] [PubMed]
- Sakurai Y, Takeda T. Acquired hemophilia A: a frequently overlooked autoimmune hemorrhagic disorder. J Immunol Res 2014;2014:320674 [Crossref] [PubMed]
Cite this article as: Melo AJ, Vadhan JD, Miller M, Melo JB. Acquired hemophilia A presenting as spontaneous hemorrhage following corpus luteum cyst rupture: a case report and review of the literature. J Emerg Crit Care Med 2021;5:33.


