
Triple threat: bilateral renal artery thrombosis and heparin induced thrombocytopenia in a patient with COVID-19, a case report
Introduction
Over 23 million Americans have tested positive for COVID-19 since the onset of the pandemic (1). As cases climb, the systemic manifestations of this virus have been unmasked. For instance, the thrombotic sequelae of COVID-19 have posed a particular challenge for clinicians. They are thought to be caused by the excessive inflammation, platelet activation, and endothelial dysfunction induced by the virus (2). It is estimated that up to 50% of patients with severe COVID-19 disease are diagnosed with coagulopathy, such as deep venous thrombosis or pulmonary emboli (3). Thus, thromboprophylaxis is strongly recommended in COVID-19 patients, leading to the widespread use of heparin in this patient population (4). Heparin-induced thrombocytopenia is the feared complication of heparin administration (5). In this immune mediated condition, platelets are consumed due to the interaction between the FcγIIa receptor and the heparin, platelet factor (PF) 4, and anti-PF4-heparin antibody complex (5). Paradoxically, heparin-induced thrombocytopenia can result in both thrombocytopenia and a prothrombotic state. Thus far, heparin-induced thrombocytopenia has been uncommonly diagnosed in COVID-19 patients (6). Here, we present a unique case of a previously healthy middle-aged man who developed heparin-induced thrombocytopenia after being treated for bilateral renal artery thrombosis as a result of his COVID-19 related complication. Amongst the millions of COVID-19 infections, less than 10 cases of bilateral renal artery thrombosis have been reported in COVID-19 patients (7). To our knowledge, this is the only recorded instance of a patient with COVID-19, heparin-induced thrombocytopenia, and bilateral renal artery thrombosis.
We present the following article in accordance with the CARE reporting checklist (available at https://dx.doi.org/10.21037/jeccm-21-41).
Case presentation
Our patient is a 44-year-old man with a significant past medical history of recurrent kidney stones who presented to an outside hospital emergency room complaining of sudden onset severe, diffuse abdominal pain associated with bilateral lower extremity numbness and tingling. He had tested positive for COVID-19 four days prior. He denied shortness of breath, cough, nausea, vomiting, diarrhea, a decrease in urine volume and loss of taste and smell. The patient did not take any medications at home on a regular basis, although he took one dose of Ibuprofen 400 mg the day of his visit to the emergency department in an attempt to control his abdominal pain. He had no history of established care with a nephrologist. He reported consuming between four to five alcoholic drinks a month and using marijuana about once a week. His family history was positive for polycystic kidney disease in his sister.
In the emergency department, the patient was afebrile, his heart rate was 89 beats/min, respiratory rate was 18 breaths/min, and his oxygen saturation was 95% on room air. His blood pressure was 156/96 mmHg upon arrival to the hospital, and rose to 190/94 mmHg one hour later. His physical exam revealed his abdomen to be soft, symmetric, non-distended, and diffusely tender to deep palpation. There were no visible lesions or scars, aorta was midline without bruit or visible pulsation, and bowel sounds were present and normoactive in all four quadrants. His exam was negative for costovertebral tenderness, psoas sign, obturator sign or Rovsing’s sign. He had trace bilateral pitting edema without overlying erythema or cellulitis. The remainder of his physical exam was negative for acute abnormalities.
The patient’s lab values revealed his creatinine to be 3.05 mg/dL, white blood cell count to be 16.5×103/µL, and platelets to be 295×103/µL. The remainder of his complete blood count, metabolic panel and coagulation studies were within normal limits. Radiograph of the chest showed diffuse bilateral infiltrates. His subsequent CT scan of his abdomen and pelvis with IV contrast showed bilateral renal artery thrombosis (Figure 1). The patient was promptly treated with a heparin drip, then transferred to our facility for bilateral renal artery angiogram and catheter directed thrombolysis with tPA. Intra-operatively, his disease was characterized as extensive, nearly occlusive bilateral renal artery thrombosis with collateral flow to the upper poles. The patient reported his abdominal pain as mildly improved after the procedure. He was admitted to the ICU, where he continued his catheter-directed thrombolysis with tPA. In the intensive care unit, the patient was placed on 4 liters nasal cannula. Treatment for COVID-19 included dexamethasone, azithromycin, ceftriaxone, and vitamin supplementations. The following day, his creatinine increased to 5.78 mg/dL. Nephrology was consulted for worsening renal function. Hypercoagulable studies of factor V activity, protein C activity, free protein S, factor V Leiden mutation, and an ANA screen were within normal limits. His echocardiogram showed normal ejection fraction without intracardiac thrombi. Lower extremity duplex was negative for deep vein thrombosis.
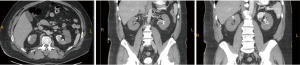
On hospital day three, the patient’s creatinine rose to 8.02 mg/dL (Table 1). His d-dimer was elevated to 21.63 mg/L. He underwent a second bilateral renal angiogram, which showed that majority of thrombi within bilateral renal arteries had been successfully treated, with persistent occlusion of some renal artery branches (Figure 2A,2B). His kidneys demonstrated minimal heterogeneous cortical enhancement, likely due to bilateral renal infarcts. Catheter-directed thrombolysis was discontinued and anticoagulation with systemic heparin was started (Figure 2A,2B). On the following day, his renal function continued to decline on medical floor, and he was initiated on hemodialysis while being monitored for signs of renal recovery.
Table 1
| Laboratory values | Admission | Day 1 | Day 5 | Day 13 |
|---|---|---|---|---|
| Creatinine (mg/dL) | 3.05 | 8.05 | 6.99 | 6.87 |
| D-Dimer (mg/L FEU) | No value | 21.63 | No value | 5.32 |
| Platelets×103/μL | 295 | 213 | 149 | 115 |

Additional laboratory studies to evaluate the patient’s hypercoagulability were negative. These included: c-ANCA, p-ANCA, atypical p-ANCA, myeloperoxidase antibody, beta-2 GPI panel, glomerular basement membrane antibody, anti-prothrombin IgG, phosphatidylserine IgG and IgM, anti-cardiolipin antibodies and complement C3. His hepatitis panel and HIV tests were negative. IgA, IgM, IgG and kappa and lambda light chain tests were negative for acute pathology, without M spike and without light chain 100:1 ratio.
Between hospital admission day 7 and day 9, the patient’s platelet counts decreased from 235×103/µL to 184×103/µL. They continued to decrease, falling to 124×103/µL on day 12, with a nadir of 115×103/µL on day 13—representing a 51.1% decrease over a five-day period. HIT antibody optical density was elevated to 1.960, confirming the diagnosis of heparin-induced thrombocytopenia. The patient’s heparin drip was discontinued, and he was transitioned to apixaban 5 mg twice a day. The patient was discharged after fifteen days of hospitalization and referred to outpatient dialysis treatment. The patient was amenable to the plan at discharge and reported profound symptomatic improvement.
Of note, all procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Discussion
Treating disease related to COVID-19 was the main battle field for healthcare workers in 2020. Our current understanding of hematological manifestations of this disease is its tendency to cause significant thrombotic phenomenon (8). In the care of our healthy 44-year-old patient, the initial challenge was obtaining the correct diagnosis in the emergency department. Given his history of kidney stones, nephrolithiasis complicated by pyelonephritis was an early working differential diagnosis. Another differential diagnosis included renal artery stenosis due to his hypertension and elevated creatinine on presentation. Computed tomography has a higher sensitivity for renal artery thrombosis as compared to renal ultrasound (9). This imaging study can also detect the presence of nephrolithiasis, a condition with similar presenting symptoms (9). The prevention of irreversible renal damage in the context of renal artery thrombosis depends on early diagnosis and the prompt initiation of medical therapy (10). Once the diagnosis of renal artery thrombosis is made, it is essential to refer the patient for emergency reperfusion where interventional radiology is available. Prior case studies have demonstrated that catheter-directed thrombolysis for acute renal artery occlusion is a safe treatment modality. It has been successful at restoring kidney function even in patients with prolonged renal ischemia (10).
The next step in the diagnostic pathway was to investigate the origin of his thrombosis. Traditionally, bilateral renal arterial thrombi are rare, and are usually caused by emboli of cardiac origin, either primary or paradoxical (11). Other conditions associated with bilateral renal artery thrombosis include trauma, sickle cell disease, antiphospholipid antibody syndrome and cocaine use (12). Once we ruled out these causes with laboratory findings, echocardiography and venous duplex, we suspect that our patient’s renal artery thrombosis was related to his COVID-19 infection were connected. Kidney disease is a known sequela of COVID-19, and renal artery thrombosis has been specifically implicated as a cause of kidney dysfunction in COVID-19 patients (13). Additionally, renal artery thrombosis is a rare but documented cause of acute abdomen (14). This confirms the association of between the patient’s acute kidney injury, COVID-19 infection and abdominal pain.
Bridging medical therapy with intravenous heparin upon completion of catheter-directed thrombolysis has demonstrated efficacy in prior instances of renal artery thrombosis (15). One of the complications associated with heparin use is heparin induced thrombocytopenia, which is most commonly linked to post-cardiac surgery patients (16). Recent data suggests that the cumulative incidence of heparin induced thrombocytopenia (HIT) antibodies in COVID-19 patients may be more than quadruple that of hospitalized but COVID-19 negative patients (17). One study found that the cumulative incidence of positive HIT immunoassay assay was 12% at 25 days (95% CI, 4% to 26%) in patients hospitalized with COVID-19 (17). Treatment of acute heparin induced thrombocytopenia involves immediate cessation of heparin, and the subsequent use of non-heparin anticoagulants, such as argatroban, bivalirudin, danaparoid, fondaparinux, and direct oral anticoagulants (18).
For our patient, while actively addressing his renal artery thrombosis, we aggressively treated his COVID-19 infection based on current available evidence. He received dexamethasone, azithromycin, ceftriaxone, and oral vitamins. The RECOVERY trial demonstrated the utility of dexamethasone 6 mg/d for up to 10 days in patients hospitalized with COVID-19 (19). A study performed in Wuhan, China found that antibiotics were associated with improved mortality in patients hospitalized with COVID-19 (20).
Conclusions
In conclusion, our patient’s presentation is another colorful tessera contributing to the mosaic of COVID-19. His COVID-19 infection, bilateral renal artery thrombosis and concurrent heparin induced thrombocytopenia emphasize the array of clinical findings that can accompany COVID-19. Given the robust association between COVID-19 and bleeding, clotting, and overall microvascular injury, it is likely that future studies will further investigate the mechanisms of these effects. Clinical vigilance remains as important as ever as the variety of clinical presentations the virus can induce unfold before us. Obtaining timely diagnosis in order to initiate life-saving treatment highlights success in battling COVID-19.
Acknowledgments
Funding: This research was supported (in whole or in part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this publication represent those of the author(s) and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://dx.doi.org/10.21037/jeccm-21-41
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at https://dx.doi.org/10.21037/jeccm-21-41). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- CDC COVID Data Tracker [Internet]. Centers for Disease Control and Prevention. Centers for Disease Control and Prevention; [cited 2021Mar14]. Available online: https://covid.cdc.gov/covid-data-tracker/
- Philipponnet C, Aniort J, Chabrot P, et al. Renal artery thrombosis induced by COVID-19. Clin Kidney J 2020;13:713. [Crossref] [PubMed]
- Miesbach W, Makris M, et al. COVID-19: Coagulopathy, Risk of Thrombosis, and the Rationale for Anticoagulation. Clin Appl Thromb Hemost 2020;26:1076029620938149. [Crossref] [PubMed]
- Lingamaneni P, Gonakoti S, Moturi K, et al. Heparin-Induced Thrombocytopenia in COVID-19. J Investig Med High Impact Case Rep 2020;8:2324709620944091. [Crossref] [PubMed]
- Huang CT, Hsu SY, Chang KW, et al. Heparin-induced thrombocytopenia and thrombosis in a patient with Covid-19. Thromb Res 2020;196:11-4. [Crossref] [PubMed]
- Bidar F, Hékimian G, Martin-Toutain I, et al. Heparin-induced thrombocytopenia in COVID-19 patients with severe acute respiratory distress syndrome requiring extracorporeal membrane oxygenation: two case reports. J Artif Organs 2021;24:277-81. [Crossref] [PubMed]
- Acharya S, Anwar S, Siddiqui FS, et al. Renal artery thrombosis in COVID-19. IDCases 2020;22:e00968. [Crossref] [PubMed]
- Iba T, Levy JH, Connors JM, et al. The unique characteristics of COVID-19 coagulopathy. Crit Care 2020;24:360. [Crossref] [PubMed]
- Arabi M, Vellody R, Cho K, et al. Acute Renal Artery Occlusion with Prolonged Renal Ischemia: A Case of Successful Treatment with Stent Placement and Catheter-directed Thrombolysis. J Clin Imaging Sci 2011;1:11. [PubMed]
- Amilineni V, Lackner DF, Morse WS, et al. Contrast-enhanced CT for acute flank pain caused by acute renal artery occlusion. AJR Am J Roentgenol 2000;174:105-6. [Crossref] [PubMed]
- El Shamy O, Munoz-Casablanca N, Coca S, et al. Bilateral Renal Artery Thrombosis in a Patient With COVID-19. Kidney Med 2021;3:116-9. [Crossref] [PubMed]
- Lopez VM, Glauser J, et al. A case of renal artery thrombosis with renal infarction. J Emerg Trauma Shock 2010;3:302. [Crossref] [PubMed]
- Sauerberg N, Khan YS. Renal Artery Thrombosis. Treasure Island (FL): StatPearls Publishing; 2021.
- Singh S, Wang L, Yao QS, et al. Spontaneous renal artery thrombosis: an unusual cause of acute abdomen. N Am J Med Sci 2014;6:234-6. [Crossref] [PubMed]
- Nandwani A, Pathania D, Jha PK, et al. Renal Artery Thrombosis with Renal Infarction: A Rare Cause of Acute Abdomen. Indian J Nephrol 2017;27:313-5. [Crossref] [PubMed]
- Warkentin TE, Greinacher A, et al. Heparin-induced thrombocytopenia and cardiac surgery. Ann Thorac Surg 2003;76:638-48. [Crossref] [PubMed]
- Patell R, Khan AM, Bogue T, et al. Heparin induced thrombocytopenia antibodies in COVID‐19. Am J Hematol 2020;95:E295-6. [Crossref] [PubMed]
- Cuker A, Arepally GM, Chong BH, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: heparin-induced thrombocytopenia. Blood Adv 2018;2:3360-92. [Crossref] [PubMed]
- RECOVERY Collaborative Group. Dexamethasone in Hospitalized Patients with Covid-19. N Engl J Med 2021;384:693-704. [Crossref] [PubMed]
- Liu C, Wen Y, Wan W, et al. Clinical characteristics and antibiotics treatment in suspected bacterial infection patients with COVID-19. Int Immunopharmacol 2021;90:107157. [Crossref] [PubMed]
Cite this article as: Benge EJ, McWhorter Y. Triple threat: bilateral renal artery thrombosis and heparin induced thrombocytopenia in a patient with COVID-19, a case report. J Emerg Crit Care Med 2022;6:3.


