Lemierre’s syndrome: a role for thrombectomy, a case report
Introduction
Lemierre syndrome is characterized by thrombophlebitis of the vasculature of the head and neck following an oropharyngeal infection. Sepsis and septic emboli are common, with emboli primarily affecting the lungs (1). The primary source of infection is commonly the palatine tonsils and peritonsillar tissue; other sources include the lungs, middle ear, mastoid, teeth, and sinuses (2). The disease was first described by Courmont and Cade in 1900 and named in 1936, when Lemierre described 20 cases of “an aerobic septicemia” with a 90% mortality rate (1,3,4). Before antibiotics, the disease was common and prognosis grim, but with increased use of penicillins, Lemierre’s syndrome became known as the “forgotten disease” (1,5-7). There has been a re-emergence, first noted in the 1990s likely due to microbiome resistance (1,2,6,7). Incidence is estimated at 0.8–3.6 per 1 million persons, and mortality has improved from 90% to 0–18% (1,8,9).
Antibiotic coverage is the cornerstone of treatment and systemic anticoagulation is also used commonly (1-7,10,11). Thrombectomy seems like a logical therapy, but its role is not well described. Thrombectomy is a commonly performed procedure in most hospitals with an interventional radiologist on staff. The process itself is minimally invasive and results in rapid to immediate symptomatic relief for the patient. A large clot burden in the internal jugular vein (IJV) can result in diminished blood flow in that vessel, increased septic emboli to the lungs as well as lead to increased airway edema from venous congestion. Interventional percutaneous procedures, like thrombectomy should be considered earlier in patients refractory to antibiotics alone. Thrombectomy could potentially decrease duration of ICU stay as well as overall hospital stay, decrease disease progression such as in the development of cavitary pulmonary septic embolic or simply just improve patient symptomology. Further studies would be required to prove the aforementioned items but randomized clinical trials may be a bleak endeavor given the rarity of this disease.
We describe the case of a 55-year-old woman who presented with altered mental status in the setting of a sore throat, whose hospital course was complicated by acute respiratory failure requiring ventilatory support and failure of antibiotic and anticoagulation therapy. It was only after thrombectomy, performed by Interventional Radiology, that she started to demonstrate significant clinical improvement with reduced airway edema that allowed for extubation. We present the following case report with accordance to the CARE reporting checklist (available at https://dx.doi.org/10.21037/jeccm-21-38).
Case presentation
A 55-year-old woman with no significant past medical history presented as a transfer from an outside hospital for otorhinolaryngology evaluation. She initially presented four days prior with a two-week history of neck pain associated with anorexia, fatigue, lethargy, and fevers. She denied smoking and illicit drug use. Patient history was noncontributory. She was febrile to 103 degrees Fahrenheit, normotensive and tachycardic to 110. She had a new oxygen requirement of 4 liters nasal cannula. Physical exam was notable for high pitched stridor. Neck exam revealed tenderness to palpation laterally but no apparent bulging masses or lymphadenopathy. Abdominal, neurological, and musculoskeletal examinations were normal.
Diagnostic investigations
Initial laboratory data is shown in Table 1. Electrocardiogram (EKG) demonstrated sinus tachycardia. Chest X-ray demonstrated no acute pulmonary disease processes. Computed tomography (CT) scans demonstrated a 1.5 cm × 3 cm retropharyngeal abscess (RPA) spanning C1-C2 associated with bilateral IJV thrombosis and patchy ground glass opacities in all lung fields.
Table 1
| Variable | Labs at first hospital | Reference range; first hospital | On admission; this hospital | At discharge this hospital | Reference range; this hospital |
|---|---|---|---|---|---|
| Blood | |||||
| Hematocrit (%) | 28.3 | 34.0–43.0 | 23.4 | 31.4 | 37.0–47.0 |
| Hemoglobin (g/dL) | 9.4 | 12.0–16.0 | 8.3 | 10.3 | 12.0–16.0 |
| White-cell count (per mm3) | 33.1 | 4.8–10.8 | 38.6 | 4.88 | 4.8–10.8 |
| Differential count (%) | |||||
| Neutrophils | 80.8 | 38–78 | 93 | 49 | 40–65 |
| Bands | 6 | 0–10 | 0 | ||
| Metamyelocytes | 0 | 0–0 | 0 | 0–0 | |
| Lymphocytes | 2.5 | 15–20 | 4 | 32 | 21–44 |
| Monocytes | 16.3 | 0–10 | 3 | 12 | 4–9 |
| Eosinophils | 0.1 | 0–7 | 6 | 0–5 | |
| Basophils | 0.3 | 0–3 | 2 | 0–2 | |
| Platelet count (per mm3) | 305 | 150–450 | 276 | 547 | 130–400 |
| Sodium (mmol/L) | 123 | 136–145 | 131 | 137 | 135–145 |
| Potassium (mmol/L) | 2.9 | 3.5–5.1 | 3.4 | 3.8 | 3.5–5.0 |
| Chloride (mmol/L) | 82 | 98–107 | 97 | 101 | 95–105 |
| Carbon dioxide (mmol/L) | 30 | 22–29 | 26 | 30 | 22–30 |
| Urea nitrogen (mg/dL) | 54 | 6–20 | 48 | 11 | 7.0–17.0 |
| Creatinine (mg/dL) | 1.6 | 0.5–0.9 | 1.2 | 0.7 | 0.7–1.2 |
| Glucose (mmol/L) | 138 | 74–109 | 124 | 86 | 75–110 |
| Lactate (mmol/L) | 1.9 | 0.5–2.2 | 1.2 | 0.5–2.2 | |
| Alanine aminotransferase (IU/L) | 62 | 10–35 | 64 | 16 | 10–45 |
| Aspartate aminotransferase (IU/L) | 116 | 10–35 | 126 | 24 | 15–50 |
| Alkaline phosphate (IU/L) | 271 | 35–104 | 231 | 63 | 40–125 |
| Total bilirubin (mg/dL) | 4.1 | 0–1.2 | 3.7 | 0.3 | 0.2–1.3 |
| Direct bilirubin (mg/dL) | 2.6 | 0–0.2 | 1.4 | 0–0.3 | |
| D-dimer (mg/L) | 2.31 | 0.19–0.49 | |||
| Pro-calcitonin (ng/mL) | 3.36 | 0–0.099 | |||
| Erythrocyte sedimentation rate (mm/h) | 102 | 88 | 0–30 | ||
| Lactate dehydrogenase (IU/L) | 624 | 389 | 300–600 |
Human immunodeficiency virus, viral respiratory panel and rapid plasma reagin (RPR) were negative. Blood and urine cultures were negative. Repeat chest X-ray demonstrated bilateral patchy opacities in the lingula, left lower lobe and right lower lobe. Given concern for airway compromise, ENT was consulted for airway examination and incision and drainage of the abscess.
Differential diagnosis
The differential diagnosis includes pneumonia, endocarditis, infectious mononucleosis, pharyngeal abscess, malignancy of the head and neck. Diagnosis is purely clinical and should be suspected in patients presenting with oropharyngeal infection lasting greater than one week with fever, oropharyngeal pain, ipsilateral neck pain/swelling, and tenderness on exam (2-5). Visualization of thrombus in the jugular veins with infiltrates in the lung confirms this diagnosis.
Treatment
Initial treatment at the outside hospital included resuscitation with fluids, antibiotics (Ceftriaxone, Vancomycin, and Metronidazole), therapeutic anticoagulation with enoxaparin, and dexamethasone for airway edema. Her course was complicated by mild hypoxia and increased oxygen requirement.
Following the transfer, the patient arrived hemodynamically stable on 4L NC. Antibiotics were broadened to piperacillin-tazobactam, enoxaparin was switched to therapeutic heparin, otorhinolaryngology was emergently consulted for evaluation. A flexible fiberoptic laryngoscope demonstrated a patent airway with mild edema and no other abnormalities. Infectious disease recommended de-escalation of antibiotic coverage to Ampicillin-Sulbactam.
On hospital day 2, the patient developed a new facial droop, acute mental status change, and dysphonia concerning for an acute stroke. CT brain demonstrated no evidence of acute processes. Later that day, she developed increased work of breathing, and an audible stridor. Repeated flexible laryngoscopy demonstrated no new changes compared to her prior exam. Given her increased work of breathing, she was trialed on high flow nasal cannula, transitioned to BiPAP, and eventually intubated for airway protection in the setting of worsening positional stridor and hypercapnia.
Antibiotic coverage was broadened to include metronidazole and vancomycin given worsening clinical status. A repeat CT with intravenous contrast demonstrated a 2.2 cm × 1.7 cm × 5.1 cm RPA that extended to the arch of C1, with inferior extension to C3-C4 vertebral bodies (Figure 1A,B). There was complete occlusion of the airway apart from the endotracheal tube, and the airway was not visualized until the level of C6-C7.

The CT demonstrated complete venous occlusion and a thrombus beginning at the right distal transverse sinus extending to the level of the external jugular vein branch confluence (Figure 2A). There was an incomplete thrombus extending from the left jugular bulb, intermittently throughout the left IJV (Figure 2B). Cavitating ground glass opacities were seen within the lungs. Given these findings, the patient was deemed a poor candidate for incision and drainage.

Interventional radiology was consulted for consideration of thrombectomy but during informed consent the family elected to continue conservative management, primarily due to lack of evidence for mechanical thrombectomy.
On hospital day 6, repeat CT scan revealed decreasing phlegmon size but increasing airway compression, and ongoing evolution and cavitation. Due to airway compression caused by venous congestion and edema, and concern for worsening septic emboli to her lungs, the decision was made to proceed with thrombectomy. A Flowtriever catheter (Inari Medical) was deployed, and a significant amount of thrombus along the R IJV was mechanically aspirated with immediate improvement in blood flow (Figure 3A,B).

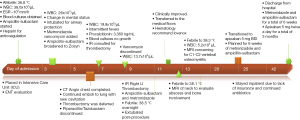
Following thrombectomy, airway edema decreased rapidly, and the patient was successfully extubated on hospital day 8. She was transitioned to enoxaparin from heparin, and antibiotics were de-escalated to ampicillin-sulbactam. She was transitioned out of the Intensive care unit on hospital day 10. A timeline of the patient’s hospitalization is presented in Figure 4 and denotes keys events during her hospitalization.
Outcome and follow-up
The patient’s remaining hospital course was significant for C1-C2 vertebral body osteomyelitis requiring long-term antibiotics. Repeat imaging of the retropharyngeal phlegmon demonstrated decreasing size. The patient was eventually discharged home with a 3-month prescription of Lovenox injections and instructions to follow-up with her PCP, ENT, infectious disease, and hematology physicians. See Figure 4 for full hospital course.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Patient perspective
The patient was relieved to be extubated and downgraded from the ICU. On discharge, she was very happy and expressed gratitude for her care but was concerned about reoccurrence despite her planned close follow-up with multiple specialties.
Discussion
Lemierre’s syndrome is seen in healthy young adults, with about 51% of cases occurring in the second decade of life (3,4). Predominance has laterality, the right or the left side, yet rarely both (1). Only a handful of cases describe bilateral thrombi (1). Sore throat is usually the first symptom (83% of cases) followed by fever (82–100%), neck pain and tenderness (52%) (1-3).
Diagnosis is based on oropharyngeal infection, IJV thrombophlebitis, persistent bacteremia, and septic emboli (3). The most common pathogen is the anaerobe Fusobacterium necrophorum, a member of the normal oropharyngeal flora (3-5), leading to pharyngitis, local invasion of the pharyngeal space, and IJV causing septic thrombophlebitis and systemic signs of infection.
Antibiotic therapy is first line treatment. The antibiotic regimen should consist of an antibiotic with a beta-lactamase inhibitor since treatment failure with penicillin has been reported (8,10). Piperacillin-tazobactam, imipenem, or ceftriaxone in conjunction with metronidazole are acceptable regimens (8). Antibiotic therapy should be initiated promptly and timely as there is a 25–30% mortality increase when antibiotics are withheld for 4 or more days (1,8,9). Given the low incidence, there is currently no data to guide therapy duration. A duration of 4 weeks, including at least 2 weeks of intravenous therapy, stands as the current recommendation (12).
Anticoagulation is debated, and it is uncertain whether it may reduce the propagation of thrombus or embolic events. Conflicting results from limited case reports demonstrate favorable outcomes with and without anticoagulation. The use of anticoagulation in the setting of progression of thrombosis into the cerebral sinuses, presence of significant clot burden, continued fever or bacteremia after five to seven days of appropriate antimicrobial therapy, or if complications of septic emboli occur is widely accepted (3,5,8-11). Opposition to anticoagulation argues that source control performed by surgical intervention plus antibiotics achieve complete resolution. Approximately, 20–30% of patients are treated with anticoagulation despite the debate (2,8). Pre-antibiotics, surgical interventions included IJV ligation or incision, which almost always improved clinical outcomes (1,3,8); now, it is reserved as a last resort for severe disease (5,8). Anticoagulation was utilized in this case, but it was not until thrombectomy that the patient’s status clinically improved.
The role of mechanical thrombectomy has not been well described, and less than a handful of case reports have been published or even mention mortality benefit. Currently, the need for interventional procedures is guided by clinical circumstances. Over the last five to seven years, more reports have emerged with the use of thrombectomy and other percutaneous mechanical antithrombotic procedures as a possible treatment options for thrombophlebitis associated with Lemierre syndrome refractory to standard medical care (11,13). This patient continued to deteriorate while receiving standard of care with antibiotics and systemic anticoagulation. It was speculated that her clot burden with her bilateral IJV thrombosis was causing venous congestion and worsening laryngopharynx edema leading to airway compromise and neurological changes. Her rapid clinical improvement post-procedure suggests that in this case, thrombectomy was an important and integral therapy.
Lemierre syndrome is a rare disease, for which the mainstay therapy has always been conservative with antibiotics and anticoagulation, but with the advancement of medical technology, new methodologies should be adopted to treat disease. In this case, mechanical thrombectomy performed in the IR suite proved invaluable in leading to reduced morbidity and length of stay. Further studies are required to prove morbidity and mortality benefit.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://dx.doi.org/10.21037/jeccm-21-38
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://dx.doi.org/10.21037/jeccm-21-38). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Moore BA, Dekle C, Werkhaven J. Bilateral Lemierre's syndrome: a case report and literature review. Ear Nose Throat J 2002;81:234-6, 238-40, 242 passim. [Crossref] [PubMed]
- Eilbert W, Singla N. Lemierre's syndrome. Int J Emerg Med 2013;6:40. [Crossref] [PubMed]
- Hagelskjaer LH, Prag J, Malczynski J, et al. Incidence and clinical epidemiology of necrobacillosis, including Lemierre's syndrome, in Denmark 1990-1995. Eur J Clin Microbiol Infect Dis 1998;17:561-5. [Crossref] [PubMed]
- Karkos PD, Asrani S, Karkos CD, et al. Lemierre's syndrome: A systematic review. Laryngoscope 2009;119:1552-9. [Crossref] [PubMed]
- Lu MD, Vasavada Z, Tanner C. Lemierre syndrome following oropharyngeal infection: a case series. J Am Board Fam Med 2009;22:79-83. [Crossref] [PubMed]
- Hagelskjaer Kristensen L, Prag J. Human necrobacillosis, with emphasis on Lemierre's syndrome. Clin Infect Dis 2000;31:524-32. [Crossref] [PubMed]
- Camelo C, Brandao M, Fernandes L, et al. Lemierre syndrome: a case report. J Vasc Bras 2015;14:253-7. [Crossref]
- Riordan T. Human infection with Fusobacterium necrophorum (Necrobacillosis), with a focus on Lemierre's syndrome. Clin Microbiol Rev 2007;20:622-59. [Crossref] [PubMed]
- Riordan T, Wilson M. Lemierre's syndrome: more than a historical curiosa. Postgrad Med J 2004;80:328-34. [Crossref] [PubMed]
- De Smet K, Claus PE, Alliet G, et al. Lemierre’s syndrome: a case study with a short review of literature. Acta Clin Belg 2019;74:206-10. [Crossref] [PubMed]
- Kar S, Webel R. Septic thrombophlebitis: percutaneous mechanical thrombectomy and thrombolytic therapies. Am J Ther 2014;21:131-6. [Crossref] [PubMed]
- Hagelskjaer Kristensen L, Prag J. Lemierre's syndrome and other disseminated Fusobacterium necrophorum infections in Denmark: a prospective epidemiological and clinical survey. Eur J Clin Microbiol Infect Dis 2008;27:779-89. [Crossref] [PubMed]
- Ashraf M, Laurich C, Huntington MK. Septic thrombophlebitis of the internal jugular and subclavian veins treated with percutaneous mechanical thrombolysis. JMM Case Reports 2015;2. [Crossref]
Cite this article as: Davis J, Adedeji O, Hawkins K, Akkari R, Seneff M. Lemierre’s syndrome: a role for thrombectomy, a case report. J Emerg Crit Care Med 2022;6:4.