
Providing background for antimicrobial stewardship strategy using costs data: a mission impossible?
Introduction
Existing health systems sustainability depends on the ability to reduce waste and costs. Among the drug-related cost, one of the most relevant is that induced by antibiotics being about one-third of hospitalized patients, and more than two-thirds of critical patients treated with them (1,2). It has also been reported that around 30% to 50% of the prescribed antibiotics are unnecessary prescribed or inappropriate (3-5), leading to an avoidable health expenditure that has been estimated to be $35.1 billion (6) in 2013 in the United States.
Antibiotic-resistant micro-organism has been reported to cause yearly, in Europe, at least 400,000 cases of antibiotic-resistant infections with 25,000 deaths and hospitalization costs higher than 1.5 billion euros (7). The need of limiting the increase of multi-drug-resistant (MDR) infections might be associated with the possibility of contain spending.
COVID-19 pandemic exacerbated the problem, forcing intensive care units (ICUs) to work in a state of emergency and increasing either the empirical antibiotic use or antimicrobial resistance (8). Other aspects of the clinical course of COVID-19 patients contributed to exacerbate critical issues, such as the difficult distinction between infectious and non-infectious causes of respiratory deterioration and the much-debated relevance of fungal and viral co-infections (9-11).
The challenge that is now involving not only high-income, but also low- and middle-income countries (12), is therefore the implementation of effective antimicrobial stewardship (AS) projects to control spending and contain antibiotic resistance growth.
Even if the literature is consistent that adherence to local guidelines, use of targeted therapies, early suspension of unnecessary therapies, shift from intravenous to oral therapy, should all to be included in AS programs (13), the general characteristics of these programs (14-16) and the measures to evaluate their effectiveness are still unclear. This is particularly true if we refer to the ICU contest, where the lack of dedicated human resources and the poor effectiveness of available measurement parameters are limiting further their implementation (17-19).
We therefore tested the possibility of providing the basis for an effective AS project to be developed in the context of the Department of Anesthesia and Intensive Care starting from the situation described by the administrative reports produced every six months by the Hospital Management Control Structure.
Methods
The Department of Anesthesia and Intensive Care of ‘Città della Salute e della Scienza’ hospital of Turin (Italy) include eight ICUs: two general, one neuro-surgical, one cardio-surgical, one obstetric-gynecological, one mainly dedicated to trauma, one pediatric and one directly located in the Emergency Department with an ICU/general beds relationship equal to 71/1,910.
Cost data referring to anti-microbial drugs used in the Department, proposed as surrogate consumption indices, were evaluated referring to the first 6 months of year 2015 and 2016, using the reports produced by the Management Control Structure. Data were analyzed in collaboration with an infectious disease specialist and all ICU prescribers were actively involved in the analysis. The ethics approval and informed consent were not required as this study focused on cost data.
Due to the fact that a subset of antimicrobial drugs is known to be particularly prescribed to treat infections induced by MDR pathogens, MDR infections trends and characteristics in five out of the eight ICUs of the Department (two general, one neuro-surgical, one cardio-surgical and one directly located in the Emergency Department) using the same Microbiological Laboratory were specifically studied.
The actual use of indirect fungal infection markers, such as 1,3-b-D-glucan, in supporting the antifungal drugs prescriptions was even evaluated.
Results
Both in 2015 or in 2016, the twelve most expensive antimicrobials induced, by their own, an expenditure equal to 783,681/1,763,084 euros (44.5%) and 697,058/1,659,702 (42%) of the total expenditure of the Department (Table 1). More detail, although the more expensive and used drugs remained in the 2-year period considered, Linezolid, Tigecycline, Daptomycin, Meropenem, Teicoplanin and Colistin (Table 1) a comparison between 2015 and 2016 shows a reduction in Tigecycline and Linezolid expenditure and a slight increase in Daptomycin and Meropenem expenditure (Figure 1).
Table 1
| Drugs | First semester 2015 | First semester 2016 | |||
|---|---|---|---|---|---|
| Cost (€) | % | Cost (€) | % | ||
| Antibiotics | 269,568 | 17.3 | 242,340 | 14.6 | |
| Linezolid | 88,625 | 5.7 | 73,849 | 4.5 | |
| Tigecycline | 106,128 | 6.8 | 69,130 | 4.2 | |
| Daptomycin | 22,555 | 1.4 | 34,576 | 2.1 | |
| Meropenem | 18,710 | 1.2 | 29,011 | 1.7 | |
| Teicoplanine | 12,605 | 0.8 | 8,965 | 0.5 | |
| Colistin | 10,609 | 0.7 | 8,000 | 0.5 | |
| Piperacillin/Tazobactam | 10,332 | 0.7 | 14,073 | 0.8 | |
| Antifungals | 514,113 | 33.0 | 454,718 | 29.2 | |
| Amphotericin B | 231,281 | 15.0 | 221,655 | 13.4 | |
| Voriconazole | 21,185 | 1.4 | 18,158 | 1.1 | |
| Echinocandins | 261,647 | 16.8 | 214,904 | 12.9 | |
| Caspofungin | 223,009 | 14.3 | 137,801 | 8.3 | |
| Anidulafungin | 33,813 | 2.2 | 75,093 | 4.5 | |
| Micafungin | 4,824 | 0.3 | 2,010 | 0.1 | |
Percentages are computed referring to the total expenditure for drugs referring to the same period.

Among antifungal drugs, representing altogether a percentage of expenditure equal to 33% and 29.2% (respectively in 2015 and 2016) of the total expenditure on antimicrobial drugs, a major role seems to be due to Amphotericin (15% vs. 13.4%, respectively in 2015 and 2016) and Echinocandins (16.8% vs. 12.9%, respectively in 2015 and 2016). It should be noticed, comparing 2015 and 2016 a small reduction of global antifungal consumption associated with an increase in the use of Anidulafungin (from 2.2% in 2015 to 4.5% in 2016).
As expected, and probably due to the different characteristics of patients admitted in the different ICUs, a huge variability in the use of antimicrobial drugs has been observed among the eight ICUs considered in the present analysis (Table 2). However, supporting the reproducibility of the data collection method used, these differences remained stable over the two considered semesters.
Table 2
| Type of ICU | 1° semester of 2015 | % on total expenditure | 1° semester of 2016 | % on total expenditure |
|---|---|---|---|---|
| General ICU 1 | 236,278 | 30 | 232,308 | 33 |
| Cardio-surgical ICU | 59,374 | 8 | 89,400 | 13 |
| General ICU 2 | 251,089 | 32 | 222,340 | 32 |
| ED-ICU | 34,544 | 4 | 20,062 | 3 |
| Neurosurgical ICU | 4,480 | 1 | 11,593 | 2 |
| Trauma ICU | 80,932 | 10 | 55,340 | 8 |
| Gynecological and obstetric ICU | 20,269 | 3 | 4,235 | 1 |
| Pediatric ICU | 56,447 | 7 | 31,225 | 4 |
| Cardio-surgical pediatric ICU | 40,268 | 5 | 30,032 | 4 |
Percentages are computed referring to the total expenditure for drugs in each ICU in the same period. ICU, intensive care unit; ED, emergency department.
MDR germs, especially Carbapenem resistant-Klebsiella pneumonia subtype KPC, Acinetobacter baumannii and extended spectrum beta-lactamase (ESBL) Escherichia coli, were higher in 2016 in comparison with 2015 (Figures 2,3), mainly in the ICUs admitting more severe patients such as general and cardio-surgical ones. For the treatment of these infections, that, according to literature, are commonly approached with Meropenem, Colistin and Tigecycline, around 6.4% of total drug-expenditure of the Department has been used.


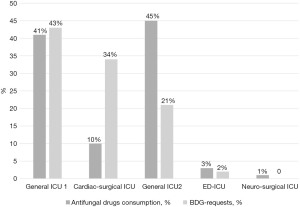
Regarding the use of 1,3-b-D-glucan to support the prescription of antifungal drugs we evidenced a diffused under-use of this diagnostic methods, with an inverse relationship with antifungal drugs prescription and 1,3-b-D-glucan use (Figure 4).
Discussion
Several projects have been proposed in the context of the ‘Città della Salute e della Scienza’ hospital with the aim of describing and optimizing the practice of antimicrobial therapy. Due to the number of antimicrobial drugs used and the lack of human resources available to collect timely data regarding on the use of these drugs, ICUs always remained outside these programs. Using the economic data routinely collected by the Management Control Structure, we were able to obtain a reliable estimate of the consumption of antimicrobial drugs in all the ICUs constituting the Department of Anesthesia and Intensive Care.
The results so far obtained confirmed that antimicrobial drugs are heavily impacting the Department’s budget, representing about 50% of the drug costs, but also that huge differences are present within the ‘intensive care’ domains.
Although some of these observed differences can be justified taking into account the different case-mix of the different ICUs of the Department, as in the case of the particularly complex transplanted patients, the results of the analysis correlating the incidence of MDR and the use of MDR-specific drugs seems to highlight a too ‘liberal’ use of high-cost drugs (Meropenem, Colistin and Tigecycline) whose use should be instead limited to ‘rescue’ therapies to apply only in the presence of MDR germs. And this evidence strongly supports the need to boost the ‘carbapenem sparing culture’, strongly recommended in the literature (21), which seems to be absent in our department. This objective, combined with the need to better analyze the differences, till now hypothesized and now demonstrated, confirmed that it worth implementing an AS program in the Department of Anesthesia and Intensive Care.
Another interesting result is that about 30% of the expenses for antimicrobial drugs are due to the purchase of antifungal drugs (mainly echinocandins and amphotericin). These drugs, in the great majority of cases, are prescribed empirically in absence of adequate diagnostic test, either direct culture, that are known to be difficult to obtain and rarely positive, or indirect testing such as 1,3-b-D-glucan whose negative predictive value has been reported (22), and recently confirmed (23).
The work has several limitations. First, cost data, chosen as readily available and part of a routine surveillance, were taken as surrogate consumption indices. However, it must be considered that an increase in in expenditure is not necessarily due to increased consumption since the total expenditure depends on both the unit cost and the frequency of use.
Second, cost data were computed using the so called ‘weighted average price’ and not the ‘definite daily dose’ (DDD) used by standard literature to estimate the consumption (24).
Third, changes in drug availability due to the introduction of new molecules or expiration of patents risk invalidating the cost analysis and must be critically evaluated by the multidisciplinary AS team.
Finally, the data presented here refer to a pre-pandemic context and therefore antecedent to the development of new important antimicrobials gradually added in recent years to the therapeutic armamentarium particularly against MDR pathogens. However, it should be noted that their prescription, at least in Italy is regulated by the infectious diseases’ specialist, and therefore already part of the AS programs.
Furthermore, even if the data collected show a reduction in the costs incurred for antimicrobials between 2015 and 2016 and an apparent increase in cases of infection with multidrug-resistant pathogens, the design of the study and the methodology used do not allow a statistical analysis that allows an interpretation of the observed phenomena.
Similarly, the relapses of a more widespread use of indirect diagnostic methods, such as the use of BDG, cannot be read as the result of the clinical implementation of an intervention. However, it should be noted that the correlation between the use of this biomarker and the reduction in the cost of antifungals is to be considered of interest for future studies designed for this purpose.
Despite the limitations illustrated above, the results so far obtained clearly support the need for a thorough review of the antimicrobial prescribing approach used up to now in the ICUs of the Department of Anesthesia and Intensive Care of the Città della Salute e della Scienza’ university hospital of Turin (Italy).
Beyond this, we do consider the ability of this approach to provide a picture of the antimicrobial practice used in the Intensive Care facilities using administrative data that are routinely collected by the Control Management Structure of each Italian hospital to monitor the achievement of the objectives annually assigned to the operating structures extremely interesting and new. Furthermore, it must be considered as an approach capable of considering costs may be useful to engage administrators for support for stewardship strategies and make them attractive and cost-effective also in terms of investments.
Even with their intrinsic limitations, these data are, in fact, always available, regardless of dedicated human resources, and therefore always potentially usable to provide the basis for an AS project aimed at continuous improvement of clinical practice and data containment.
Acknowledgments
The authors thank all the health care providers and collaborators working in the ICUs in ‘Città della Salute e della Scienza’ Hospital (Turin, Italy) for their precious and continuous collaboration and support.
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Journal of Emergency and Critical Care Medicine for the series “Risk Factors and Management of Hospital Acquired Infections in ICU”. The article has undergone external peer review.
Data Sharing Statement: Available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-27/dss
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-27/coif). The series “Risk Factors and Management of Hospital Acquired Infections in ICU” was commissioned by the editorial office without any funding or sponsorship. GM served as the unpaid Guest Editor of the series. GM received payments for lectures from GILEAD, AMBU, PFIZER, TERMOFISHER and for participation to Advisory Board from GILEAD. SC received payments for lectures from GILEAD and PFIZER. AC received payments for lectures from GILEAD and AMBU. LB received payments for lectures and presentations from GILEAD, AMBU, PFIZER, THERMOFISHER, GETTING and for participation to Advisory Board from GILEAD and 3M. The authors have no other conflicts of interest to declare.
Ethical Statement:
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Vincent JL, Rello J, Marshall J, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA 2009;302:2323-9. [Crossref] [PubMed]
- Fridkin SK, Steward CD, Edwards JR, et al. Surveillance of antimicrobial use and antimicrobial resistance in United States hospitals: project ICARE phase 2. Project Intensive Care Antimicrobial Resistance Epidemiology (ICARE) hospitals. Clin Infect Dis 1999;29:245-52. [Crossref] [PubMed]
- Leung E, Weil DE, Raviglione M, et al. The WHO policy package to combat antimicrobial resistance. Bull World Health Organ 2011;89:390-2. [Crossref] [PubMed]
- Available online: https://www.cdc.gov/drugresistance/threat-report-2013/pdf/ar-threats-2013-508.pdf
- National action plan for combating antibiotic-resistant bacteria. [cited 2017 Dec 12]. Available online: https://www.cdc.gov/drugresistance/pdf/national_action_plan_for_combating_antibotic-resistant_bacteria.pdf
- Avoidable Costs in Healthcare. [cited 2017 Dec 12]. Available online: http://www.drugstorenews.com/sites/drugstorenews.com/files/Avoidable%20Costs%20in%20Healthcare.pdf
- Healthcare associated infections antimicrobial use PPS. Available online: https://ecdc.europa.eu/sites/portal/files/media/en/publications/Publications/healthcare-associated-infections-antimicrobial-use-PPS.pdf
- Montrucchio G, Corcione S, Sales G, et al. Carbapenem-resistant Klebsiella pneumoniae in ICU-admitted COVID-19 patients: Keep an eye on the ball. J Glob Antimicrob Resist 2020;23:398-400. [Crossref] [PubMed]
- Greene MH, Nesbitt WJ, Nelson GE. Antimicrobial stewardship staffing: How much is enough? Infect Control Hosp Epidemiol 2020;41:102-12. [Crossref] [PubMed]
- Schouten J, De Waele J, Lanckohr C, et al. Antimicrobial stewardship in the ICU in COVID-19 times: the known unknowns. Int J Antimicrob Agents 2021;58:106409. [Crossref] [PubMed]
- Montrucchio G, Lupia T, Lombardo D, et al. Risk factors for invasive aspergillosis in ICU patients with COVID-19: current insights and new key elements. Ann Intensive Care 2021;11:136. [Crossref] [PubMed]
- Pierce J, Apisarnthanarak A, Schellack N, et al. Global Antimicrobial Stewardship with a Focus on Low- and Middle-Income Countries. Int J Infect Dis 2020;96:621-9. [Crossref] [PubMed]
- Barlam TF, Cosgrove SE, Abbo LM, et al. Implementing an Antibiotic Stewardship Program: Guidelines by the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America. Clin Infect Dis 2016;62:e51-77. [Crossref] [PubMed]
- Schuts EC, Hulscher MEJL, Mouton JW, et al. Current evidence on hospital antimicrobial stewardship objectives: a systematic review and meta-analysis. Lancet Infect Dis 2016;16:847-56. [Crossref] [PubMed]
- Filice G, Drekonja D, Greer N, et al. Antimicrobial Stewardship Programs in Inpatient Settings: A Systematic Review. Washington (DC): Department of Veterans Affairs (USA); 2013. (VA Evidence-based Synthesis Program Reports). Available online: http://www.ncbi.nlm.nih.gov/books/NBK253513/
- Davey P, Brown E, Charani E, et al. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst Rev 2013;CD003543. [PubMed]
- Pulcini C, Morel CM, Tacconelli E, et al. Human resources estimates and funding for antibiotic stewardship teams are urgently needed. Clin Microbiol Infect 2017;23:785-7. [Crossref] [PubMed]
- Karanika S, Paudel S, Grigoras C, et al. Systematic Review and Meta-analysis of Clinical and Economic Outcomes from the Implementation of Hospital-Based Antimicrobial Stewardship Programs. Antimicrob Agents Chemother 2016;60:4840-52. [Crossref] [PubMed]
- Montrucchio G, Sales G, Corcione S, et al. Choosing wisely: what is the actual role of antimicrobial stewardship in Intensive Care Units? Minerva Anestesiol 2019;85:71-82. [Crossref] [PubMed]
- Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect 2012;18:268-81. [Crossref] [PubMed]
- Palacios-Baena ZR, Gutiérrez-Gutiérrez B, Calbo E, et al. Empiric Therapy With Carbapenem-Sparing Regimens for Bloodstream Infections due to Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae: Results From the INCREMENT Cohort. Clin Infect Dis 2017;65:1615-23. [Crossref] [PubMed]
- Posteraro B, Tumbarello M, De Pascale G, et al. (1,3)-β-d-Glucan-based antifungal treatment in critically ill adults at high risk of candidaemia: an observational study. J Antimicrob Chemother 2016;71:2262-9. [Crossref] [PubMed]
- Pappas PG, Kauffman CA, Andes DR, et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis 2016;62:e1-50. [Crossref] [PubMed]
- WHOCC. WHO Collaborating Centre for Drug Statistics Methodology. Definition and general considerations. Available online: https://www.whocc.no/ddd/definition_and_general_considera/
Cite this article as: Montrucchio G, Sales G, Corcione S, Curtoni A, Urbino R, Brazzi L. Providing background for antimicrobial stewardship strategy using costs data: a mission impossible? J Emerg Crit Care Med 2022;6:21.


