
Diagnostic and therapeutic considerations in idiopathic systemic capillary leak syndrome: a case report
Introduction
Systemic capillary leak syndrome (SCLS) was first described by Bayard Clarkson in 1960 (1). It is characterised by increased capillary permeability leading to severe shock, hemoconcentration, hypoalbuminemia. The disease is rare and under recognized. Whereas in other forms of shock a restrictive fluid balance might be beneficial, in SCLS resuscitation remains the most important aspect of treatment. In this case reports we therefore describe the main diagnostic considerations of SCLS and discuss therapeutic pitfalls. We present the following case in accordance with the CARE reporting checklist (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-22/rc).
Case presentation
A 46-year-old female patient with a medical history of core rod myopathy, cholecystectomy and hypothyroidism visited the emergency department twice in a period of two months. Written informed consent for publishing this report was obtained. First, she presented because of diffuse muscle weakness (legs more than arms) and a mild shortness of breath for a week. In the emergency department she was hypotensive (78/50 mmHg), anuric and collapsed several times. Laboratory results demonstrated a markedly elevated hematocrit (0.59 L/L), leukocytosis (19.6×109/L, mostly neutrophil granulocytes, without lymphocytosis, lymphopenia or eosinophilia), mildly decreased kidney function and a slightly elevated lactate (2.7 mmol/L). Despite resuscitation with 8 L of fluid (NaCl 0.9%), the patient remained hypotensive. She was admitted to the medium care unit for vasopressor therapy (norepinephrine). The patient developed a normal anion gap (hyperchloremic) metabolic acidosis and after another three litres of fluids (NaHCO3 1.4% and Ringer’s lactate) vasopressors were stopped. Within 24 h the patient was discharged to the internal medicine ward and kidney function recovered completely. She was discharged six days after admission. Since there were no signs of cardiac dysfunction on echocardiography, no pulmonary embolism on CT-angiography, no sepsis or anaphylaxis, this episode of shock was diagnosed as hypovolemic due to mild nausea, vomiting and diarrhoea. At that time, no other explanation could be found. One month later, she visited the emergency department with similar symptoms. Besides diffuse weakness and collapse there was a mild cough. She had tested positive for COVID-19 just two days before (despite being fully vaccinated). On examination there was hypotension (90/70 mmHg), mild tachycardia (110/min), anuria and mild tachypnoea (23 breaths per minute) without other respiratory problems. Despite aggressive fluid resuscitation with 12 L of fluids and additional vasopressor therapy, the patient remained hemodynamically unstable and laboratory results revealed hemoconcentration (haematocrit from 0.45 to 0.59 L/L) lactic acidosis and hypoalbuminemia (Figure 1, Table 1). The patient was admitted to the ICU. Due to the large amount of fluids administered pulmonary edema was likely to occur in the following days. Therefore, the patient was intubated to prevent clinical deterioration due to pulmonary edema. Within the next 24 h, the cumulative positive fluid balance increased to 38 L (crystalloids and NaHCO3 1.4%). For a short period of time high doses of vasopressors (noradrenaline 0.5 µg/kg/min and vasopressin 0.04 IE/min) were given as well, without effect on blood pressure.
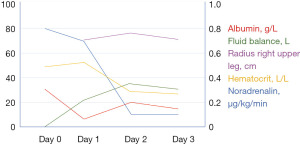
Table 1
| Test | Day 0 | Day 1 | Day 2 | Day 3 |
|---|---|---|---|---|
| Albumin (g/L) | 29 | 6 | 20 | 16 |
| Hematocrit (L/L) | 0.48 | 0.59 | 0.32 | 0.26 |
| Fluid balance (L) | 0.1 | 21 | 35 | 32 |
| Radius right upper leg (cm) | − | 69 | 74 | 71 |
| Noradrenalin (µg/kg/min) | 0.8 | 0.7 | 0.1 | 0.1 |
| Platelet count | 302 | 144 | 87 | 71 |
Laboratory results showed no signs of adrenal insufficiency, no clues for systemic mastocytosis or hereditary angioedema or hypothyroidism (Table 2).
Table 2
| Laboratory results on admission | Result [reference] |
|---|---|
| Complement component 4 (mg/L) | 123 [100–400] |
| Immunoglobulin G (g/L) | 6.37 [7–16] |
| Immunoglobulin A (g/L) | 1.43 [0.7–4] |
| Immunoglobulin M (g/L) | 1.17 [0.4–2.3] |
| Total protein (g/L) | 35 [60–80] |
| Myeloma protein (g/L) | 2.7 |
| Tryptase (μg/L) | 2.2 [<11.4] |
| Thyroid-stimulating hormone (mE/L) | 2.54 [0.26–4.2] |
| Cortisol (μmol/L) | 0.75 [0.19–0.55] |
After ruling out cardiogenic, distributive and obstructive forms of shock, we diagnosed the patient with SCLS. Besides typical haemoconcentration, hypoalbuminemia and clinical capillary hyperpermeability, we additionally detected a IgG type κ Myeloma protein (M-protein), which is associated with SCLS.
Next, treatment with intravenous immunoglobulins (IVIG) (1 g/kg) was initiated and twenty-four hours later, hemodynamics stabilized.
Fluid administration was stopped and because of the highly positive fluid balance and its potential severe complications, diuretics were given and continuous renal replacement therapy (CRRT) was started to remove excessive fluids. Nonetheless, the ICU stay was complicated by development of compartment syndromes in both arms and legs with creatinine kinase (CK) levels up to 46,000 U/L, for which fasciotomy was performed four days after admission. Our patient had decreasing platelet levels which might have been a clue for disseminated intravascular coagulation (DIC) and although our patient had the clinical signs of a compartment syndrome, limb ischemia due to DIC could not be excluded. In the end we concluded that the presence of muscle necrosis of the left leg was due to a combination of compartment syndrome, the high dose of vasopressor agents used and possibly DIC. In the recovery phase the patient remained on renal replacement therapy for a couple of weeks, but in the end her kidney function recovered to an estimated clearance of 30 mL/min. Her hospital stay was prolonged due to infected muscle necrosis, eventually leading to amputation of both legs. After approximately 2.5 months of admission, the patient was discharged for rehabilitation. In the rehabilitation clinic IVIG is given subcutaneous every four weeks.
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Discussion
Idiopathic SCLS, also called Clarkson’s disease, was first described by Bayard Clarkson in 1960 (1). He characterised the syndrome as recurrent episodes of shock with haemoconcentration, increased capillary permeability leading to edema and eventually organ failure. The aetiology of the disease was unclear, however, he did find abnormal gamma globulins and described recurrent viral infections prior to the episodes of shock. Nowadays, SCLS is characterised by a prodromal phase (this phase may be absent in idiopathic SCLS), a leak phase and a recovery phase. In the prodromal phase there is a trigger for the systemic leakage. This is most frequently a viral infection, however also stem cell transplantation, anti-cancer treatment [for example SS1P, IL-1, IL-2, anti-CD22 (2), GM-CSF and checkpoint inhibitors (3-5)], physical exertion and snake bites can induce severe systemic capillary leakage (6). Since the COVID-19 pandemic, relapses of SCLS (7), as well as new onset SCLS have been reported (8). As for our patient, we did not find a clear trigger for the first episode, whereas the second episode could have been triggered by a COVID-19 infection.
The pathophysiology of SCLS includes an increase in capillary permeability to medium sized proteins and plasma. As a consequence, proteins and fluid leak from the intravascular into the interstitial space, resulting in hypotension, edema and hypoalbumenia. The larger red blood cells remain intravascular, resulting in marked hemoconcentration. In most cases this leads to hypovolemic shock. The increase in capillary permeability is thought to be due to cytokines released by various stimuli. These cytokines might disrupt adherend junctions between cells, increasing vascular permeability even more. However, studies at a cellular level are scarce. One study demonstrated elevated vascular endothelial growth factor (VEGF), which is involved in regulating adherence junctions, and angiopoietin-2 (Ang2) levels in SCLS patients (9). Besides cytokines, up to 90% of SCLS patients have a monoclonal gammopathy of unknown significance (MGUS), as already described by Clarkson (10).
Despite the clinical characteristics often seen in SCLS, there are no strict criteria to diagnose this syndrome. Literature suggests the following clinical characteristics and laboratory findings support the diagnosis of SCLS (Table 3).
Table 3
| Major | Minor |
|---|---|
| Hemoconcentration (despite fluid resuscitation) | Elevated VEGF |
| Hypovolemic shock | Elevated angiopoietin 2 |
| Hypoalbuminemia | Present M-protein/MGUS |
| Generalised edema | |
| Other forms of shock ruled out |
SCLS, systemic capillary leak syndrome; VEGF, vascular endothelial growth factor; M-protein, myeloma protein; MGUS, monoclonal gammopathy of unknown significance.
The corner stone in the treatment of SCLS is fluid resuscitation. This is different from several other forms of shock where a restrictive fluid management and a more liberal use of vasopressors seems beneficial. SCLS patients have severe hypovolemia and the use of vasopressor agents without adequate fluid therapy may be detrimental. The choice of vasopressor therapy is important as well, as vasopressin proved to give more peripheral muscle necrosis (11). Besides frequent laboratory measurements (haematocrit, albumin, lactate), echocardiography can be used to monitor the effect of resuscitation. Furthermore minimal-invasive monitoring, such as pulse contour analysis, or invasive monitoring, for example with a Swan-Ganz catheter, can be used to evaluate fluid responsiveness. Intubation should be considered early in the disease to avoid a difficult intubation because of oedema in the pharynx and larynx. To avoid complications due to excessive fluid administration, it is important to initiate diuretic treatment or renal replacement therapy as soon as vascular hyperpermeability decreases. Besides symptomatic treatment with fluids, no other effective treatments are known. In several case reports a potential benefit of IVIG is described (12). However, their mechanism of action remains unknown and a benefit on survival due to IVIG treatment could not be found (13). Besides, IVIG is associated with acute kidney injury. In our patient clinical improvement may be due to IVIG treatment or the natural course of the disease. The acute kidney injury in our patient already occurred before starting IVIG treatment. However, it may have been a contributing factor.
Since several patients also had elevated VEGF levels, additional treatment with bevacizumab was also proposed. Bevacizumab, used in the treatment of several forms of cancer, acts by selectively binding circulating VEGF, thereby inhibiting the binding of VEGF to its cell surface receptors. This inhibition leads to a reduction in microvascular growth of tumor blood vessels and thus limits the blood supply to tumor tissues. Since adherence junctions dissolve in response to VEGF (14), possibly aggravating capillary leak, it was postulated that bevacizumab could possibly diminish this phenomenon. Last year, two case reports described successful treatment with bevacizumab. The first patient was a young female suffering from idiopathic capillary leak syndrome (CLS), resembling our patient. Her treatment included IVIG on days 3–4, a subsequently bevacizumab on day 5 (15). The second patient was an infant after stem cell transplantation who developed CLS with response one day after administration of bevacizumab (16). Conversely, another report showed no benefit of bevacizumab in the context of CLS after stem cell transplantation in a 58-year-old woman (17). This may be due to the different pathogenesis of SCLS in children and adults. In murine studies canagliflozin was able to prevent vascular hyperpermeability, and thus might be a hopeful new therapeutic agent (18). It remains to be elucidated to what extent recovery can be attributed to bevacizumab, earlier interventions including IVIG, or the natural course of CLS. However, any shortening of the leakage phase could attenuate the risk of complications. Adjunct treatment with bevacizumab could be considered, further research however is warranted. We advocate a treatment regimen with mainly resuscitation, early intubation to prevent difficult intubation due to laryngeal edema, IVIG in persistent shock, CRRT when necessary and early use of diuretics when shock reverses (Figure 2).
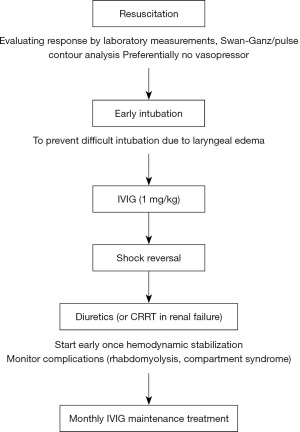
Complications in the disease course of SCLS may be due to the disease itself or because of the treatment. Both hypotension and volume depletion may lead to acute organ failure whereas high dose of vasopressors (in combination with hypovolemia) may lead to ischemia and worsen organ failure. Moreover, IVIG is associated with renal failure. Because of the large amounts of fluids given, compartment syndrome of all extremities is also frequently seen (12). Compartment syndrome may even be a presenting symptom of SCLS (19).
Idiopathic SCLS is a recurrent syndrome. In patients with recurrent episodes of SCLS, β2-agonists (e.g., theophylline) or monthly treatment with IVIG (1–2 g/kg) prevented attacks in about half of the patients and could therefore be beneficial (20). No other preventive treatment has proven beneficial yet. Possibly there is a role for bevacizumab in the future, but further research is necessary.
In conclusion, SCLS is a rare disorder that is characterised by hypovolemic shock with hemoconcentration despite resuscitation, hypoalbuminemia, and progressive organ failure. This diagnose needs to be considered in patients in which hemoconcentration occurs despite extensive fluid resuscitation. Since vasopressor use may aggravate ischemic complications in a hypovolemic condition its use is discouraged in these patients. In addition, the use of IVIG might be beneficial. Whether the use of bevacizumab is also of value is yet unclear.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-22/rc
Peer Review File: Available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-22/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-22/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Clarkson B, Thompson D, Horwith M, et al. Cyclic edema and shock due to in-creased capillary permeability. Amer J Med 1960;29:193-216. [Crossref] [PubMed]
- Jeong GH, Lee KH, Lee IR, et al. Incidence of Capillary Leak Syndrome as an Adverse Effect of Drugs in Cancer Patients: A Systematic Review and Meta-Analysis. J Clin Med 2019;8:143. [Crossref] [PubMed]
- Lescure C, Lescoat A, Salé A, et al. Systemic Capillary Leak Syndrome (Clarkson's Disease) as a Complication of Anti-Programmed Death 1 Immunotherapy. J Thorac Oncol 2019;14:e131-2. [Crossref] [PubMed]
- Polishchuk I, Yakobson A, Zemel M, et al. Nivolumab-induced systemic capillary leak syndrome as an ultra rare life-threatening phenomenon of late toxicity and intravenous immunoglobulin efficacy. Immunotherapy 2021;13:807-11. [Crossref] [PubMed]
- Percik R, Nethanel A, Liel Y. Capillary-leak syndrome: an unrecognized early immune adverse effect of checkpoint-inhibitors treatment. Immunotherapy 2021;13:653-9. [Crossref] [PubMed]
- Siddall E, Khatri M, Radhakrishnan J. Capillary leak syndrome: etiologies, pathophysiology, and management. Kidney Int 2017;92:37-46. [Crossref] [PubMed]
- Pineton de Chambrun M, Cohen-Aubart F, Donker DW, et al. SARS-CoV-2 Induces Acute and Refractory Relapse of Systemic Capillary Leak Syndrome (Clarkson's Disease). Am J Med 2020;133:e663-4. [Crossref] [PubMed]
- Knox DB, Lee V, Leither L, et al. New-Onset Systemic Capillary Leak Syndrome in an Adult Patient with COVID-19. Case Rep Crit Care 2021;2021:8098942. [Crossref] [PubMed]
- Xie Z, Ghosh CC, Patel R, et al. Vascular endothelial hyperpermeability induces the clinical symptoms of Clarkson disease (the systemic capillary leak syndrome). Blood 2012;119:4321-32. [Crossref] [PubMed]
- Atkinson JP, Waldmann TA, Stein SF, et al. Systemic capillary leak syndrome and monoclonal IgG gammopathy; studies in a sixth patient and a review of the literature. Medicine (Baltimore) 1977;56:225-39. [Crossref] [PubMed]
- Ozel Coskun BD, Karaman A, Gorkem H, et al. Terlipressin-induced ischemic skin necrosis: a rare association. Am J Case Rep 2014;15:476-9. [Crossref] [PubMed]
- Lambert M, Launay D, Hachulla E, et al. High-dose intravenous immunoglobulins dramatically reverse systemic capillary leak syndrome. Crit Care Med 2008;36:2184-7. [Crossref] [PubMed]
- Pineton de Chambrun M, Luyt CE, Beloncle F, et al. The Clinical Picture of Severe Systemic Capillary-Leak Syndrome Episodes Requiring ICU Admission. Crit Care Med 2017;45:1216-23. [Crossref] [PubMed]
- Claesson-Welsh L. Vascular permeability--the essentials. Ups J Med Sci 2015;120:135-43. [Crossref] [PubMed]
- Kouadri G, Perzo N, Sauvetre G, et al. Refractory severe idiopathic systemic capillary leak syndrome successfully treated with bevacizumab: a case report. Angiogenesis 2021;24:399-401. [Crossref] [PubMed]
- Yabe H, Yabe M, Koike T, et al. Rapid improvement of life-threatening capillary leak syndrome after stem cell transplantation by bevacizumab. Blood 2010;115:2723-4. [Crossref] [PubMed]
- Wang P, Chan CH, Chen HF, et al. Refractory systemic capillary leak syndrome treated with bevacizumab: a case report. Angiogenesis 2022;25:9-11. [Crossref] [PubMed]
- Angé M, De Poortere J, Ginion A, et al. Canagliflozin protects against sepsis capillary leak syndrome by activating endothelial α1AMPK. Sci Rep 2021;11:13700. [Crossref] [PubMed]
- Tanabe M, Hikone M, Sugiyama K, et al. Systemic capillary leak syndrome requiring fasciotomy for limb compartment syndrome: A case report and literature review. Acute Med Surg 2022;9:e756. [Crossref] [PubMed]
- Marra AM, Gigante A, Rosato E. Intravenous immunoglobulin in systemic capillary leak syndrome: a case report and review of literature. Expert Rev Clin Immunol 2014;10:349-52. [Crossref] [PubMed]
Cite this article as: Houterman M, Ellenbroek D, Humalda JK, van der Hoeven JG, Ramakers BP. Diagnostic and therapeutic considerations in idiopathic systemic capillary leak syndrome: a case report. J Emerg Crit Care Med 2022;6:26.


