
Low-dose lipid emulsion for pediatric vasoplegic shock due to quetiapine and fluvoxamine overdose: a case report
Introduction
Intravenous lipid emulsion (ILE) therapy was originally proposed for treatment of local anesthetic systemic toxicity (1). More recently, ILE has also been used to treat severe toxicity of numerous lipophilic substances (2-5). However, there are currently no dosing recommendations for toxicity of medications excluding local anesthetics; several case reports have documented successful resuscitation of such overdoses using much lower dosages of ILE than are used for local anesthetic toxicity (5). Pediatric ILE dosing is a weight-based proportion of the adult dose; as such, there is a paucity of research guiding recommendations of ILE therapy dosing in pediatric cases of lipophilic drug overdose (6). As suicide attempts via ingestion of selective serotonin reuptake inhibitor (SSRI) and/or antipsychotics, especially among adolescents, are becoming increasingly common in the United States (7), further research into ILE administration for lipophilic medication toxicity in pediatric patients is warranted. We present a case of a 17-year-old male in severe vasoplegic shock and subsequent multiorgan failure from an intentional quetiapine and fluvoxamine overdose who was successfully resuscitated with low-dose ILE therapy. We present the following article in accordance with the CARE reporting checklist (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-53/rc).
Case presentation
A 17-year-old male with a history of depression was found unresponsive 14 hours after a suicide attempt where he ingested 130 mg/kg quetiapine and 32 mg/kg fluvoxamine. He presented to an outlying emergency department (ED) where initial vitals were: Glasgow coma scale (GCS) 3, heart rate (HR) 140 beats/min, blood pressure (BP) 80/40 mmHg. First arterial blood gas (ABG) revealed a respiratory acidosis of pH 7.16/PCO2 94 mmHg/PaO2 300 mmHg/HCO3 33.5 mEq/L/base excess +1.6 mEq/L, with an elevated lactate of 7.8 mmol/L. He was intubated and central venous access obtained. He received 4 L normal saline, activated charcoal, 4 amps of sodium bicarbonate, and started on a norepinephrine infusion prior to pediatric intensive care unit (PICU) transport. On PICU arrival, patient continued to have GCS 3, mean arterial pressure (MAP) 53 mmHg and HR 154 beats/min. His initial electrocardiogram (EKG) revealed prolonged QTc of 537 milliseconds (HR 145 beats/min) with narrow QRS. Despite additional fluid resuscitation, a sodium bicarbonate infusion, and escalation of vasoactive infusions to 29 µg/min of norepinephrine, 11 µg/min of epinephrine and 16 mU/min of vasopressin, he remained in vasoplegic shock, with an ABG of pH 7.28/PCO2 41 mmHg/PaO2 77 mmHg/HCO3 19.3 mEq/L/base deficit −7.1 mEq/L, with rising lactate of 8.9 mmol/L, and MAP 66 mmHg.
Dialysis and extracorporeal membrane oxygenation (ECMO) were precluded as therapeutic options given the lipophilic nature of the ingested medications and the severity of vasoplegia, respectively. ILE therapy was discussed with toxicology experts and implemented by hour 5 of PICU admission. A shortage of lipid emulsion supply at the hospital necessitated the use of lower doses than current poison control recommendations. Standard recommendations include a 20% lipid emulsion solution administered as a 1.5 mL/kg bolus over 1 minute, followed by a 0.25 mL/kg/min continuous infusion. The patient received a 1 mL/kg bolus over 1 minute followed by 0.05 mL/kg/min continuous infusion for 2 hours (i.e., 67% and 20% of the current dosing recommendations, respectively).
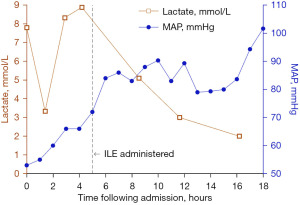
Hemodynamic improvement was noted within 30 minutes of starting the ILE therapy. MAP increased by 10 mmHg after the initial ILE bolus. The patient’s hemodynamics continued to steadily improve, and within 2 hours of ILE initiation, patient had a MAP of 84 mmHg. ABG 7 hours following ILE initiation showed improving shock with pH 7.37/PCO2 36 mmHg/PaO2 106 mmHg/HCO3 20.8 mEq/L/base deficit −3.9 mEq/L, lactate 3.0 mmol/L (Figure 1). Within 24 hours of initiating ILE therapy, the sodium bicarbonate infusion was stopped, and epinephrine and vasopressin were weaned off. By hospital day 3, norepinephrine was discontinued. For 19 hours after ILE therapy was administered, serum electrolytes could not be obtained secondary to the lipemic nature of the blood. Once the blood’s lipemic nature improved, the initial chemistries obtained revealed a severe rhabdomyolysis (peak creatinine kinase 448,929 U/L on hospital day 2). The drug-induced rhabdomyolysis resulted in acute tubular necrosis (ATN) and subsequent oliguric-anuric acute kidney injury (AKI) warranting hemodialysis (HD) initiation on hospital day 2. He received daily HD treatment from hospital day 2–6 and then was weaned to three times weekly HD sessions from hospital day 7–21 [peak serum creatinine (sCr) 10.9 mg/dL, blood urea nitrogen (BUN) 113 mg/dL on hospital day 11]. The patient was extubated on hospital day 5 and returned to full neurological baseline. His urine output spontaneously returned on hospital day 19, and he was weaned off HD support by hospital day 21. He was discharged to inpatient psychiatry on hospital day 41 with stage 3 chronic kidney disease (CKD) (sCr 1.13 mg/dL, BUN 16 mg/dL). At the patient’s most recent outpatient nephrology follow up, he demonstrated continued renal recovery to stage 2 CKD (sCr 1.01 mg/dL, BUN 11 mg/dL).
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient’s legal guardian for publication of this case report. A copy of the written consent is available for review by the editorial office of this journal.
Discussion
Given the high prevalence of depression the United States, suicide attempts with intentional overdoses of SSRIs and atypical antipsychotics are becoming increasingly common. SSRI overdose frequency has increased 20-fold in the last decade, with a 30% increase in adolescent overdoses in the past 3 years (over 10,000 cases per year) (7). Although SSRI toxicity most frequently presents with symptoms of serotonin syndrome including high fever, seizures, mydriasis, and unconsciousness, overdose may also cause cardiotoxicity via inhibition of multiple myocardial ion channels, resulting in sinus bradycardia, QT, or QRS prolongation (8). While atypical antipsychotic overdoses are less frequently reported (over 3,000 cases per year), they are associated with much higher mortality rates and are also cardiotoxic (7). Antipsychotic cardiotoxicity is mediated by alpha-1-adrenergic antagonism, resulting in vasoplegia and reflex tachycardia, as well as tachycardia due to antimuscarinic activity (9).
The mechanism by which ILE therapy treats lipophilic drug toxicity is likely multifactorial (10,11). The most historically prominent theory is that of the lipid sink, in which fat-soluble compounds are redistributed away from target organs into the intravascular compartment (1). However, a growing body of evidence supports the contribution of other mechanisms, which include the provision of cardiomyocytes with sufficient free fatty acids to use as an alternate energy source, a direct positive inotropic effect, modulation of calcium and sodium channels, and reduction of vasoplegia through inhibition of endothelial nitric oxide synthase (12-17).
While ILE therapy was originally proposed for local anesthetic systemic toxicity, over the last decade, it has been successfully used to treat severe toxicity of numerous lipophilic substances (1-5). It is currently recommended as an adjunct treatment for overdoses of tricyclic antidepressants, bupropion, calcium channel blockers, beta blockers, cocaine, diphenhydramine, and lamotrigine, as well as for life-threatening toxicity of other lipophilic drugs when other interventions fail (3). There are numerous reports documenting the use of ILE in acute second-generation antipsychotic toxicity in both adult and pediatric patients (2,4,5). While severe SSRI overdoses less frequently result in life threatening toxicity in isolation, ILE therapy has been used to treat toxicity of SSRIs in combination with antipsychotics or other psychotropic medications (2,5).
In addition to vasoplegic shock, our patient’s PICU course was complicated by oliguric-anuric AKI secondary to severe rhabdomyolysis and resulting ATN that required 19 days of HD support. Antipsychotics are indeed among the leading causes of drug-induced rhabdomyolysis (18). One case series found a 17% prevalence of myotoxicity in cases of olanzapine overdose, with creatine kinase values peaking 12 hours after ingestion (19). Earlier detection of our patient’s rhabdomyolysis and AKI was challenged by an inability to obtain lab results. For 19 hours after ILE administration, serum blood sent to the laboratory could not yield electrolyte results due to processing error caused by lipemia. Once chemistries resulted, it corroborated severe rhabdomyolysis and AKI. There was discussion among the intensive care unit (ICU) team whether the patient’s renal morbidity could have been prevented and/or mitigated with earlier detection by serum chemistry studies. However, the sequelae of rhabdomyolysis with AKI requiring HD support was likely inevitable given the degree of the initial toxic ingestion. Addressing the patient’s unstable hemodynamics with ILE administration was the critical clinical priority over ability to monitor labs. In fact, by the patient receiving a lower dose of the ILE therapy, it is plausible that the lipemia lasted a shorter overall duration, minimizing the time that serum chemistries could not be obtained.
Common adverse effects of ILE therapy include hypertriglyceridemia, pancreatitis, and phlebitis, were not experienced in this case (10). The patient did experience lipemia, which is an inherent consequence of ILE administration. The ICU team was unable to obtain accurate serum chemistry labs for 19 hours following the ILE infusion. The mechanisms by which lipemia causes an inability to obtain serum laboratories are several. Lipemic blood causes physical, chemical, and spectrophotometric method interferences, non-homogeneity in the serum sample, and a volume displacement effect between the aqueous and lipid phases (20). All of these factors result in lab processing error and explain why after ILE administration, serum electrolyte laboratories are commonly unobtainable until resolution of lipemia.
The current recommended ILE dosing regimen in the treatment of lipophilic drug overdose by toxicology experts consists of a 1.5 mL/kg lean body mass of 20% lipid emulsion bolus, followed by a continuous infusion of 0.25 mL/kg/min, with options to repeat the bolus or double the infusion rate for persistent cardiovascular instability (6). Because of a limited hospital supply of lipid emulsion, the patient only received 67% of the recommended bolus and only 20% of the recommended continuous infusion rate. There is a paucity of studies describing the effect of ILE therapy in lower doses, particularly in pediatrics. One case report documented successful treatment of quetiapine and citalopram overdose in an adolescent using the recommended bolus dosing followed by a lower infusion dose of 0.025 mL/kg/min over 1 hour (5).
This case highlights the successful use of low-dose ILE therapy for SSRI and antipsychotic drug toxicity and also underscores the importance of early initiation. Our experience suggests there may be a lower dosing range for ILE therapy, both in regard to the current recommended bolus and infusion dosing, that is equally efficacious as current recommendations for the treatment of pediatric vasoplegic shock secondary to antipsychotic and SSRI toxicity. Moreover, using lower doses of ILE therapy may confer additional benefits of less pronounced and/or shorter duration of ILE side effects of lipemia and reduce overall healthcare costs. More research regarding the safe and appropriate dosing ranges of ILE therapy in children is needed.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-53/rc
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-53/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient’s legal guardian for publication of this case report. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Weinberg GL. Pretreatment or resuscitation with a lipid infusion shifts the dose-response to bupivacaine-induced asystole in rats. Anesthesiology 1998;88:1071-5. [Crossref] [PubMed]
- Eren Cevik S, Tasyurek T, Guneysel O. Intralipid emulsion treatment as an antidote in lipophilic drug intoxications. Am J Emerg Med 2014;32:1103-8. [Crossref] [PubMed]
- Gosselin S, Hoegberg LC, Hoffman RS, et al. Evidence-based recommendations on the use of intravenous lipid emulsion therapy in poisoning. Clin Toxicol (Phila) 2016;54:899-923. [Crossref] [PubMed]
- Elgazzar FM, Elgohary MS, Basiouny SM, et al. Intravenous lipid emulsion as an adjuvant therapy of acute clozapine poisoning. Hum Exp Toxicol 2021;40:1053-63. [Crossref] [PubMed]
- Purg D, Markota A, Grenc D, et al. Low-dose intravenous lipid emulsion for the treatment of severe quetiapine and citalopram poisoning. Arh Hig Rada Toksikol 2016;67:164-6. [Crossref] [PubMed]
- Nedialkov AM, Umadhay T, Valdes JA, et al. Intravenous Fat Emulsion for Treatment of Local Anesthetic Systemic Toxicity: Best Practice and Review of the Literature. AANA J 2018;86:290-7. [PubMed]
- Gummin DD, Mowry JB, Beuhler MC, et al. 2019 Annual Report of the American Association of Poison Control Centers' National Poison Data System (NPDS): 37th Annual Report. Clin Toxicol (Phila) 2020;58:1360-541. [Crossref] [PubMed]
- Pacher P, Kecskemeti V. Cardiovascular side effects of new antidepressants and antipsychotics: new drugs, old concerns? Curr Pharm Des 2004;10:2463-75. [Crossref] [PubMed]
- Stoner SC. Management of serious cardiac adverse effects of antipsychotic medications. Ment Health Clin 2018;7:246-54. [Crossref] [PubMed]
- Karcioglu O. Use of lipid emulsion therapy in local anesthetic overdose. Saudi Med J 2017;38:985-93. [Crossref] [PubMed]
- Rothschild L, Bern S, Oswald S, et al. Intravenous lipid emulsion in clinical toxicology. Scand J Trauma Resusc Emerg Med 2010;18:51. [Crossref] [PubMed]
- Kuo I, Akpa BS. Validity of the lipid sink as a mechanism for the reversal of local anesthetic systemic toxicity: a physiologically based pharmacokinetic model study. Anesthesiology 2013;118:1350-61. [Crossref] [PubMed]
- Partownavid P, Umar S, Li J, et al. Fatty-acid oxidation and calcium homeostasis are involved in the rescue of bupivacaine-induced cardiotoxicity by lipid emulsion in rats. Crit Care Med 2012;40:2431-7. [Crossref] [PubMed]
- Stehr SN, Ziegeler JC, Pexa A, et al. The effects of lipid infusion on myocardial function and bioenergetics in l-bupivacaine toxicity in the isolated rat heart. Anesth Analg 2007;104:186-92. [Crossref] [PubMed]
- Huang JM, Xian H, Bacaner M. Long-chain fatty acids activate calcium channels in ventricular myocytes. Proc Natl Acad Sci U S A 1992;89:6452-6. [Crossref] [PubMed]
- Mottram AR, Valdivia CR, Makielski JC. Fatty acids antagonize bupivacaine-induced I(Na) blockade. Clin Toxicol (Phila) 2011;49:729-33. [Crossref] [PubMed]
- Ok SH, Sohn JT, Baik JS, et al. Lipid emulsion reverses Levobupivacaine-induced responses in isolated rat aortic vessels. Anesthesiology 2011;114:293-301. [Crossref] [PubMed]
- Mousavi SR, Vahabzadeh M, Mahdizadeh A, et al. Rhabdomyolysis in 114 patients with acute poisonings. J Res Med Sci 2015;20:239-43. [PubMed]
- Waring WS, Wrate J, Bateman DN. Olanzapine overdose is associated with acute muscle toxicity. Hum Exp Toxicol 2006;25:735-40. [Crossref] [PubMed]
- Nikolac N. Lipemia: causes, interference mechanisms, detection and management. Biochem Med (Zagreb) 2014;24:57-67. [Crossref] [PubMed]
Cite this article as: Hubbard AM, House LM, Lee JM. Low-dose lipid emulsion for pediatric vasoplegic shock due to quetiapine and fluvoxamine overdose: a case report. J Emerg Crit Care Med 2022;6:28.


