
Endobronchial fibrin sealant and autologous blood patch for treatment of persistent air leak in a mechanically ventilated patient: a case report
Introduction
Alveolar-pleural fistulas (APF) are pathologic communications between the pulmonary parenchyma and the pleural space which can lead to persistent air leaks (PALs). PALs are most commonly defined as a leak lasting >5–7 days, based on expected resolution of air leak post-lobectomy (1). Others define a leak from secondary spontaneous pneumothorax lasting more than 2 days as a PAL (2,3). PALs increase the risk of infection, length of hospitalization, and hospital cost (4,5). Supportive treatments for PAL in mechanically ventilated patients include chest tube drainage to allow for pleural apposition, mechanical ventilation strategies to decrease flow across the fistula, and expedient yet thoughtful liberation from mechanical ventilation (6).
For patients who fail supportive management and are not surgical candidates, endobronchial therapies are an option. Endobronchial valves (EBVs) are one-way valves that prevent airflow distal to the valve while allowing movement of secretions and air proximally. Use of EBVs for PAL can require multiple valves and a repeat bronchoscopy is needed for removal of the valves weeks after insertion (7). Other implantable devices include silicone spigots which occlude the airway and fully covered stents which obstruct airway defects in the setting of bronchopleural fistulas (BPFs) (8,9). Multiple endobronchially instilled solutions have treated PALs, including autologous blood patches, cyanoacrylate compounds, fibrin sealants, and hydrogels (10-13).
We describe a successful case of endobronchial autologous blood patch and fibrin sealant use to treat a PAL in a mechanically ventilated patient. We present the following case in accordance with the CARE reporting checklist (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-47/rc).
Case presentation
A 61-year-old female with diabetes and no prior pulmonary disease was admitted to the intensive care unit (ICU) for necrotizing pneumonia requiring invasive mechanical ventilation. She had initially been started on bilevel positive airway pressure (BPAP) ventilation but had worsening respiratory acidosis and thus was intubated. Microbiology of endotracheal sputum was positive for Pseudomonas aeruginosa, methicillin-resistant Staphylococcus aureus, and Proteus mirabilis. She was empirically treated for pneumonia with piperacillin-tazobactam and azithromycin, then switched to ciprofloxacin and vancomycin after sensitivities returned. Her necrotizing pneumonia resulted in an APF and pneumothorax (shown in Figure 1).
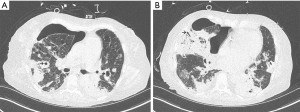
Her initial pneumothorax, discovered 4 days into her ICU admission, was large enough to cause mediastinal shift. She had an 8 French chest tube inserted and attached to suction at −20 cmH2O which led to near-complete resolution of her pneumothorax. Later that day she became hypotensive and was found to have expansion of her pneumothorax; thus, a 14 French chest tube was inserted. Despite this she did not have full re-expansion of her right lung.
After 2 days, Thoracic surgery was consulted and did not feel she was a surgical candidate given her frailty, hemodynamic instability (requiring 2 vasoactive medications), and the bilateral nature of her necrotizing pneumonia. They recommended insertion of a 24 F chest tube and to apply a suction of –40 cmH2O to all chest tubes. This led to a nearly complete re-expansion of her lung, however her air leak persisted [Cerfolio classification 4(C)].
Having failed conservative management of her PAL, she underwent an endobronchial autologous blood patch 3 days after her pneumothorax developed. Endobronchial management was attempted rather than chemical pleurodesis for two reasons: on imaging there was a culprit airway subsegment and the risk of causing acute respiratory distress syndrome (ARDS) with chemical pleurodesis in a patient actively suffering from respiratory failure was unacceptable. EBVs were not available at our institution. The patient was mechanically ventilated on pressure control ventilation with an inspiratory pressure of 22 cmH2O and a positive end expiratory pressure of 8 cmH2O. A sequential balloon occlusion technique was utilized to locate the PAL; this involved using an endobronchial balloon catheter (Disposable balloon catheter B5-2C, Olympus Medical Systems, Tokyo, Japan) to occlude the segmental airways until a decrease or resolution of the leak was achieved. The leak terminated upon occlusion of the medial segment of the right middle lobe, suggesting no significant collateral ventilation. Next, we used 25 mL of blood drawn from a central venous catheter and instilled it through the port in the endobronchial balloon catheter. We allowed the balloon to occlude for 5 minutes to allow clot formation. There was no air leak after the blood patch and 2 days later her chest tubes were removed from suction. Unfortunately, 4 days after her endobronchial blood patch she had a recurrence of her leak.
On day 8 of having a PAL, a second endobronchial blood patch was performed with the same technique as the first, however blood was instilled into both the right middle lobe medial and lateral segments and the balloon occluded the airway for 10 minutes. There was no air leak after the procedure and the chest tubes were not re-connected to suction post-procedure. The subsequent day she had an intermittent air leak [Cerfolio classification 3(I)] through her 24 French chest tube but none through the 8 French and 14 French chest tubes which were removed. Her remaining chest tube was connected to a suction of −20 cmH2O. When her chest tube was temporarily disconnected from suction for imaging tests, her pneumothorax reaccumulated and thus was reattached to suction. She underwent a tracheostomy on day 11 of her PAL.
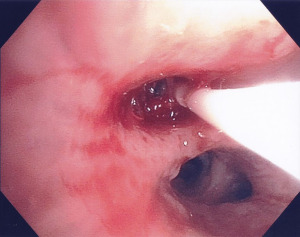
On day 15 of her PAL, she underwent a third attempt at endobronchial therapy to treat her PAL while on pressure control ventilation with an inspiratory pressure of 10 cmH2O and a positive end expiratory pressure of 5 cmH2O. Once again, 25 mL of autologous blood was instilled through the lumen of the balloon catheter and balloon occlusion was applied for 5 minutes. The next 2 mL of Tisseel fibrin sealant (Baxter Healthcare Corporation, Illinois, USA) was instilled through the balloon lumen and occlusion was applied for another 5 minutes (shown in Figure 2) as it was felt this would allow for improved airway occlusion and facilitate clot formation when compared to using autologous blood independently. She had no air leak at the end of the procedure and her chest tube was removed from suction. She tolerated the procedure well with no increase to her ventilatory support nor oxygen requirements. There was no recurrence of the air leak and her surgical chest tube was removed 23 days after her initial pneumothorax was identified. Her respiratory status improved and she no longer required invasive mechanical ventilation 32 days after her initial pneumothorax. Her air leak has not recurred to date, 4 months after her PAL.
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Discussion
Endobronchial therapies, while considered a third line treatment after conservative and surgical management, are effective in managing PALs (14,15). Although many different endobronchial therapies have been described, few modalities have been successful in mechanically ventilated patients. The most studied endobronchial treatments are one-way EBVs (7,16,17). In these case series, the number of EBVs placed ranged from 1 to 11. Deterrents to the use of valves include cost, complications (valve migration, formation of granulation tissue) (18), and the need for a repeat procedure to remove the valve.
Treating PAL in patients on mechanical ventilation is challenging and a variety of therapies have been tried. Martin et al. (19) reported a case of a patient with a BPF secondary to a necrotizing pneumonia that was treated by endobronchial instillation of tetracycline and an autologous blood patch which resulted in airway sclerosis. Another technique that has been successful for BPF closure in a mechanically ventilated patient was the combined use of a fibrin sealant and decalcified human spongiosa which was applied directly to the fistula (20).
Wiaterek et al. (21) describe a case where an absorbable hemostat (surgical) and autologous blood were used to treat an APF in a ventilated patient. In our patient we used autologous blood and a fibrin sealant which are also self-absorbable materials. A similar technique was utilized by Zhang et al. (22) where autologous blood and thrombin were used for PALs in non-ventilated patients; they reported success rates of 82% which was comparable to the 84% success rates of patients treated with silicone spigots for their PALs. They reported complications of chest pain, cough, fever, and temporary hemoptysis which occurred at similar rates for patients treated with spigots versus endobronchial autologous blood and thrombin.
Varying volumes of endobronchial autologous blood have been used in case reports with a range of 3 mL up to 100 mL (10,21-24). Generally, lower volumes have been used in cases where the blood is combined with other agents such as an absorbable hemostat or thrombin. In a case series of 4 patients successfully treated with endobronchial autologous blood patches alone, they used a volume of 100 mL (10). In a large case series where 41 patients were successfully treated with endobronchial blood and thrombin, 20–30 mL of blood was used (22).
Compared to valves, the upfront cost of absorbable materials (such as blood, fibrin sealants, and absorbable hemostat) is more economical, especially considering some patients require multiple valves for cessation of their air leaks (25,26). Additionally, since they are self-absorbable materials, they leave no lasting effect on the airways and do not require a repeat bronchoscopy for retrieval. However, one must consider that instilled endobronchial therapies may not be successful with the first attempt and require subsequent bronchoscopies for ongoing therapy. While instilled endobronchial solutions are not as easily reversible compared to endobronchial devices, should a complication occur in the setting of endobronchial blood patches with or without fibrin sealants where the clot cannot be suctioned, one could consider endobronchial instillation of tissue plasminogen activator which has been used successfully for endobronchial clot management (27).
Our case demonstrates an additional endobronchial technique to manage a PAL due to APF from a necrotizing pneumonia in a mechanically ventilated patient. The combined use of an autologous blood patch and fibrin sealant technique where balloon occlusion was maintained for 5 minutes is a safe, readily available, and inexpensive option. This technique does not require a follow-up bronchoscopy for removal of equipment and has no permanent effect on the airways.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-47/rc
Peer Review File: Available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-47/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-22-47/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Mueller MR, Marzluf BA. The anticipation and management of air leaks and residual spaces post lung resection. J Thorac Dis 2014;6:271-84. [PubMed]
- MacDuff A, Arnold A, Harvey J, et al. Management of spontaneous pneumothorax: British Thoracic Society Pleural Disease Guideline 2010. Thorax 2010;65:ii18-31. [Crossref] [PubMed]
- Mathur R, Cullen J, Kinnear WJ, et al. Time course of resolution of persistent air leak in spontaneous pneumothorax. Respir Med 1995;89:129-32. [Crossref] [PubMed]
- Liberman M, Muzikansky A, Wright CD, et al. Incidence and risk factors of persistent air leak after major pulmonary resection and use of chemical pleurodesis. Ann Thorac Surg 2010;89:891-7; discussion 897-8. [Crossref] [PubMed]
- Varela G, Jiménez MF, Novoa N, et al. Estimating hospital costs attributable to prolonged air leak in pulmonary lobectomy. Eur J Cardiothorac Surg 2005;27:329-33. [Crossref] [PubMed]
- Kempainen RR, Pierson DJ. Persistent air leaks in patients receiving mechanical ventilation. Semin Respir Crit Care Med 2001;22:675-84. [Crossref] [PubMed]
- Kalatoudis H, Nikhil M, Zeid F, et al. Bronchopleural Fistula Resolution with Endobronchial Valve Placement and Liberation from Mechanical Ventilation in Acute Respiratory Distress Syndrome: A Case Series. Case Rep Crit Care 2017;2017:3092457. [Crossref] [PubMed]
- Watanabe Y, Matsuo K, Tamaoki A, et al. Bronchial Occlusion with Endobronchial Watanabe Spigot. J Bronchol 2003;10:264-7. [Crossref]
- Cao M, Zhu Q, Wang W, et al. Clinical Application of Fully Covered Self-Expandable Metal Stents in the Treatment of Bronchial Fistula. Thorac Cardiovasc Surg 2016;64:533-9. [Crossref] [PubMed]
- Sadeghi HA. Endobronchial autologous blood patch for treatment of persistent air leak. A simple and accessible technique to treat peripheral broncho-pleural fistula. In: Interventional pulmonology [Internet]. European Respiratory Society; 2020:1203. [cited 2022 Jul 9]. Available online: http://erj.ersjournals.com/lookup/doi/10.1183/13993003.congress-2020.1203
- Scappaticci E, Ardissone F, Ruffini E, et al. Postoperative bronchopleural fistula: endoscopic closure in 12 patients. Ann Thorac Surg 1994;57:119-22. [Crossref] [PubMed]
- Hollaus PH, Lax F, Janakiev D, et al. Endoscopic treatment of postoperative bronchopleural fistula: experience with 45 cases. Ann Thorac Surg 1998;66:923-7. [Crossref] [PubMed]
- Mehta HJ, Malhotra P, Begnaud A, et al. Treatment of alveolar-pleural fistula with endobronchial application of synthetic hydrogel. Chest 2015;147:695-9. [Crossref] [PubMed]
- Dugan KC, Laxmanan B, Murgu S, et al. Management of Persistent Air Leaks. Chest 2017;152:417-23. [Crossref] [PubMed]
- Sakata KK, Reisenauer JS, Kern RM, et al. Persistent air leak - review. Respir Med 2018;137:213-8. [Crossref] [PubMed]
- Mahajan AK, Verhoef P, Patel SB, et al. Intrabronchial valves: a case series describing a minimally invasive approach to bronchopleural fistulas in medical intensive care unit patients. J Bronchology Interv Pulmonol 2012;19:137-41. [Crossref] [PubMed]
- Flora AS, Aragaki-Nakahodo A, Benzaquen S. Endobronchial Valve Placement for Persistent Air Leaks Secondary to Pulmonary Infections. J Bronchology Interv Pulmonol 2021;28:47-52. [Crossref] [PubMed]
- Sciurba FC, Ernst A, Herth FJ, et al. A randomized study of endobronchial valves for advanced emphysema. N Engl J Med 2010;363:1233-44. [Crossref] [PubMed]
- Martin WR, Siefkin AD, Allen R. Closure of a bronchopleural fistula with bronchoscopic instillation of tetracycline. Chest 1991;99:1040-2. [Crossref] [PubMed]
- Baumann WR, Ulmer JL, Ambrose PG, et al. Closure of a bronchopleural fistula using decalcified human spongiosa and a fibrin sealant. Ann Thorac Surg 1997;64:230-3. [Crossref] [PubMed]
- Wiaterek G, Lee H, Malhotra R, et al. Bronchoscopic blood patch for treatment of persistent alveolar-pleural fistula. J Bronchology Interv Pulmonol 2013;20:171-4. [Crossref] [PubMed]
- Zhang HT, Xie YH, Gu X, et al. Management of Persistent Air Leaks Using Endobronchial Autologous Blood Patch and Spigot Occlusion: A Multicentre Randomized Controlled Trial in China. Respiration 2019;97:436-43. [Crossref] [PubMed]
- Mizumori Y, Nakahara Y, Kawamura T, et al. Intrabronchial Infusion of Autologous Blood Plus Thrombin for Intractable Pneumothorax After Bronchial Occlusion Using Silicon Spigots: A Case Series of 9 Patients With Emphysema. J Bronchology Interv Pulmonol 2016;23:199-203. [Crossref] [PubMed]
- Lan RS, Lee CH, Tsai YH, et al. Fiberoptic bronchial blockade in a small bronchopleural fistula. Chest 1987;92:944-6. [Crossref] [PubMed]
- Santini M, Fiorelli A, Vicidomini G, et al. Iatrogenic air leak successfully treated by bronchoscopic placement of unidirectional endobronchial valves. Ann Thorac Surg 2010;89:2007-10. [Crossref] [PubMed]
- Spotnitz WD. Fibrin Sealant: The Only Approved Hemostat, Sealant, and Adhesive-a Laboratory and Clinical Perspective. ISRN Surg 2014;2014:203943. [Crossref] [PubMed]
- Landa E, Farra W, Ehtesham M. Endobronchial tissue plasminogen activator (TPA) administration to resolve endobronchial obstruction due to significant blood clot burden in a SAR COV 2 positive patient on extracorporeal membrane oxygenation (ECMO). Chest 2021;160:A674. [Crossref]
Cite this article as: Wang M, Buchanan B, Li P. Endobronchial fibrin sealant and autologous blood patch for treatment of persistent air leak in a mechanically ventilated patient: a case report. J Emerg Crit Care Med 2022;6:29.


