
Pericarditis in the immunocompromised patient: a case report
Highlight box
Key findings
• Pericarditis can very rarely occur in neutropenic patients, and emergency physicians should be aware of this uncommon diagnosis. Clinicians should not let bias and rare associations exclude pathologies that fit the clinical presentation.
What is known and what is new?
• Neutropenia is thought to preclude the possibility of inflammatory processes, like pericarditis. Consequently, pericarditis has rarely been associated with chemotherapy or multiple myeloma and is extremely uncommon in neutropenic patients.
• Our report is among the first to describe pericarditis in the setting of neutropenia. We highlight an extremely rare connection between pericarditis, multiple myeloma, and chemotherapeutic regimens.
What is the implication, and what should change now?
• Emergency physicians should not prematurely dismiss the possibility of pericarditis in immunocompromised patients. Serial electrocardiograms may help physicians confirm this rare diagnosis.
Introduction
Pericarditis is the inflammation of the pericardium, the fibroelastic sac made up of visceral and parietal layers separated by a potential space that surrounds the heart. Pericarditis is the most common disease of the pericardium with one study from an Italian urban area suggesting an incidence of 27.7 cases per 100,000 people (1). Fifty-five percent of pericarditis cases have an unknown, or idiopathic, etiology (2). Other etiologies include viral infection, neoplasm, autoimmune, bacterial infection, and cardiac injury; tuberculosis is another common cause of effusive pericarditis, especially in Africa (1-3). Viral pericarditis usually, but not always, results in a low volume serous pericardial effusion. Meanwhile, neoplasm related pericarditis results in larger volume effusions that can be exudative, hemorrhagic, or leukocyte filled. Therefore, tamponade and a subsequent constrictive pericarditis can result from delayed diagnosis (4). Cancers are a known cause of pericarditis with the most common ones being breast cancer, lung cancer, Hodgkin’s disease, leukemia, and lymphoma by local invasion (1). However, little data exists associating multiple myeloma (MM) with pericarditis. Previous cases have linked MM with pericardial effusions (5), but reports of pericarditis without effusion in this context remain rare (6).
While radiation therapy is associated with the development of pericarditis, few case reports exist describing pericarditis subsequent to chemotherapeutic regimens. One prior case report describes acute pericarditis attributed to docetaxel, cisplatin, and 5-fluorouracil (DCF) chemotherapy regimen, which includes docetaxel, cisplatin, and 5-flourouracil (7). Likewise, Katayama et al. described a fatal case of fulminant fatal cardiotoxicity following cyclophosphamide therapy (8). Post-mortem evaluation showed myocardial hemorrhage, pericardial effusion, and fibrinous pericarditis. There were no documented cases of KDP-PACE (carfilzomib, dexamethasone, prednisone, cisplatin, doxorubicin, cyclophosphamide, and etoposide) associated with pericarditis. We present the first reported case of pericarditis following KDP-PACE therapy for MM in accordance with the CARE reporting checklist (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-23-120/rc). This reporting standard for case reports ensures that all pertinent information, including patient demographics, diagnostic assessments, therapeutic interventions, and a discussion of relevant literature is included for readers.
Case presentation
A 32-year-old man presented to the emergency department (ED) with one hour of moderate substernal chest pain that worsened with inspiration. He reported an abrupt onset of pain, which radiated to his right shoulder and was followed by diaphoresis. Pain occurred at rest and was unrelated to exertion. He had a history of MM, which was being treated with KDP-PACE. The last infusion occurred six days prior to presentation. The patient lived in the United Arab Emirates and traveled intermittently to the United States for chemotherapy treatment. Upon initial physical exam, the patient was noted to be a thin, diaphoretic male with the following vital signs: blood pressure 112/73 mmHg, heart rate 84 beats per minute, temperature 98.4 ℉, respiratory rate 18 breaths per minute, and SpO2 99% on room air. A cardiovascular exam showed normal heart rate, regular rhythm, normal heart sounds and intact distal pulses. He had no gallop, friction rub, or murmurs. He was afebrile in the ED but reported a fever at home that was treated with an acetaminophen prior to arrival. Initial labs demonstrated neutropenia with a white blood cell count of 0.4×103/µL (normal: 4,500–11,000/µL) and 17% neutrophils [absolute neutrophil count (ANC) 0.0710×103/µL] (normal: 2,000–8,250/µL), undetectable troponin (<0.010 ng/mL) (normal: <0.04 ng/mL), and a creatinine of 1.31 mg/dL (patient’s baseline: 0.91 mg/dL; normal: 0.6–1.2 mg/dL). An electrocardiogram (ECG) showed subtle diffuse ST elevation in multiple leads without reciprocal depression (see Figure 1).
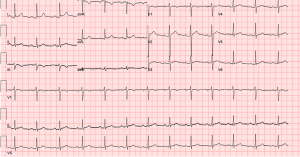
With the history of recent long-distance travel, pleuritic chest pain, and malignancy, pulmonary embolism (PE) was considered likely. Contrast computed tomography of the chest was ordered. However, the patient, who was diagnosed with acute kidney injury three months prior to presentation, declined out of concern for contrast-induced nephropathy. By that time, the patient’s creatinine had increased to 1.47 mg/dL (normal: 0.6–1.2 mg/dL). After conferring with the patient’s primary oncologist and considering his acute kidney injury, the emergency physician ordered a D-dimer test. The patient’s D-dimer was 300 µg/L (normal: 240–500 µg/L), below the threshold of concern for PE. Cardiac point-of-care ultrasound (POCUS) was obtained to evaluate for pericardial effusion. POCUS was remarkable for normal left ventricular function with absent pericardial effusion. When the patient adopted a supine position for this procedure, he reported a substantial increase in pain intensity and requested to sit up. A repeat ECG was obtained, which then showed clear ST elevation in leads I, II, aVL, aVF, V2–V6, diffuse PR depression, PR elevation in aVR, and Spodick’s sign (a downward sloping TP segment) (see Figure 2) (9).
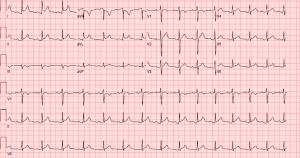
The patient was admitted to oncology, and cardiology was consulted for inpatient co-management of pericarditis. Given the patient’s recent history of acute kidney injury, the cardiologist started colchicine upon the patient’s admission rather than non-steroidal anti-inflammatory drugs (NSAIDs). On hospital day two, the physician discontinued colchicine due to a risk of adverse hematologic effects and instead initiated methylprednisolone. The patient’s cardiology consult exam noted a mild friction rub at this time. The patient was transitioned to prednisone on hospital day three, discharged with complete resolution of symptoms on hospital day four, and instructed to continue prednisone monotherapy.
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent for publication of this case report and accompanying images was not obtained from the patient or the relatives after all possible attempts were made.
Discussion
Pericarditis in the setting of neutropenia is not well-documented. There are few reported cases of this association on literature review (10), although clozapine is a known cause of both pericarditis and neutropenia. To our knowledge, this is the one of the first reported cases of a patient diagnosed with pericarditis in the setting of neutropenic fever and MM. Pericarditis has not been commonly observed in MM patients, despite its association with other malignancies (1). One possibility is that our patient developed both fever and pericarditis due to a viral infection. The majority of pericarditis cases are idiopathic and attributed to a viral source. Our patient recently received the KDP-PACE chemotherapy regimen, which is not currently associated with pericarditis. There have been some reports associating doxorubicin and cyclophosphamide with pericarditis, but these are rare (6,8). On initial consultation with the admitting oncology service, pericarditis was thought to be unlikely based on the presumed inability to mount an inflammatory response in the setting of significant neutropenia. Neutrophils are typically considered as an important part of the inflammatory pathophysiology that produces pericarditis (11). However, importantly, neutropenia does necessarily preclude the development of pericarditis, as there are many other key molecular regulators that can drive this inflammatory process such as the NLRP3 inflammasome, interleukin (IL)-1α, and IL-1β. The diagnosis of pericarditis was made when placing the patient in a supine position for bedside POCUS provoked worsening symptoms and prompted a repeat ECG with findings pathognomonic for pericarditis. A previous experimental study found that position change may induce clinically significant ECG changes, particularly among males with cardiac disease (12). Treatment options were limited for this patient due to his recent history of acute kidney injury. First line pericarditis treatments are typically aspirin or NSAIDs. Colchicine can be added when a viral etiology is suspected to improve remission rates and prevent recurrence. Steroids are the recommended second line treatment when NSAIDs are contraindicated and were ultimately used to treat this patient (13). However, a significant detriment of steroid therapy for pericarditis is a higher risk of post-treatment disease recurrence.
This case is important for emergency physicians for two primary reasons. First, the diagnosis of pericarditis was initially overlooked in favor of PE due to the patient’s recent travel, pleuritic chest pain, and malignancy; additionally, the patient’s neutropenia was thought to preclude the possibility of this inflammatory process. This bias was supported by the consulting oncologist who also conveyed a low concern for pericarditis. However, the emergency physician confirmed a diagnosis of pericarditis after noting worsening of pain with supine positioning and upon evaluation of a second ECG. Diagnostic guidelines for pericarditis generally require that patients exhibit two of the four following symptoms: pleuritic pain, ECG changes, pericardial friction rub, and pericardial effusion. Thus, the lack of an effusion or auscultated friction rub were not required for diagnosis, as our patient clinically demonstrated the first two symptoms. Emergency physicians should therefore not let bias and rare associations exclude pathologies that fit the clinical presentation. Recognition of this rare yet serious condition is of the utmost importance for emergency physicians. The diagnosis made in the ED carries significant diagnostic momentum and shapes the early inpatient stay. Additionally, if the initial diagnosis is missed, the patient may develop either a growing pericardial effusion and tamponade physiology or may develop constrictive pericarditis—both serious complications of a delayed diagnosis (4). Second, this case highlights the importance of serial ECGs in the ED. The initial ECG showed some changes but was not pathognomonic for pericarditis. Only once the emergency physician ordered a second ECG at the time of a change in systems was he able to make this rare diagnosis. Follow-up cardiology consultation confirmed the diagnosis and enabled a personalized plan of care for this patient.
Conclusions
A patient was diagnosed with acute pericarditis after presenting with neutropenic fever and a history of primary MM, which was recently treated with KDP-PACE. Like most pericarditis cases, the cause of the patient’s pericarditis was idiopathic as it is unclear whether the patient’s chemotherapy regimen or undetected viral infection was the source of inflammation. It is important not to prematurely dismiss the possibility of an inflammatory condition like pericarditis in the setting of chemotherapy-induced neutropenia. Serial ECGs may help confirm certain rare diagnoses in the ED.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-23-120/rc
Peer Review File: Available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-23-120/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jeccm.amegroups.com/article/view/10.21037/jeccm-23-120/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent for publication of this case report and accompanying images was not obtained from the patient or the relatives after all possible attempts were made.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Adler Y, Charron P, Imazio M, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC)Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2015;36:2921-64. [Crossref] [PubMed]
- Gouriet F, Levy PY, Casalta JP, et al. Etiology of Pericarditis in a Prospective Cohort of 1162 Cases. Am J Med 2015;128:784.e1-8. [Crossref] [PubMed]
- Noubiap JJ, Agbor VN, Ndoadoumgue AL, et al. Epidemiology of pericardial diseases in Africa: a systematic scoping review. Heart 2019;105:180-8. [Crossref] [PubMed]
- Tingle LE, Molina D, Calvert CW. Acute pericarditis. Am Fam Physician 2007;76:1509-14. [PubMed]
- Skipina TM, Sane DC, Cui C, et al. A plasma cell-based pericardial effusion leading to tamponade in a patient with multiple myeloma - a case report and review of the literature. Cardiovasc Pathol 2019;40:41-6. [Crossref] [PubMed]
- Bock J, Doenitz A, Andreesen R, et al. Pericarditis after high-dose chemotherapy: more frequent than expected? Onkologie 2006;29:321-4. [PubMed]
- Inanç M, Akpek M, Inanç MT, et al. Acute pericarditis during 5-fluorouracil, docetaxel and cisplatin therapy. Turk Kardiyol Dern Ars 2012;40:532-5. [Crossref] [PubMed]
- Katayama M, Imai Y, Hashimoto H, et al. Fulminant fatal cardiotoxicity following cyclophosphamide therapy. J Cardiol 2009;54:330-4. [Crossref] [PubMed]
- Chaubey VK, Chhabra L. Spodick's sign: a helpful electrocardiographic clue to the diagnosis of acute pericarditis. Perm J 2014;18:e122. [Crossref] [PubMed]
- Simão Raimundo D, Cordeiro AI, Parente Freixo J, et al. Case Report: Patient with deficiency of ADA2 presenting leukocytoclastic vasculitis and pericarditis during infliximab treatment. Front Pediatr 2023;11:1200401. [Crossref] [PubMed]
- Bonaventura A, Montecucco F. Inflammation and pericarditis: Are neutrophils actors behind the scenes? J Cell Physiol 2019;234:5390-8. [Crossref] [PubMed]
- Adams MG, Drew BJ. Body position effects on the ECG: implication for ischemia monitoring. J Electrocardiol 1997;30:285-91. [Crossref] [PubMed]
- Imazio M, Gaita F, LeWinter M. Evaluation and Treatment of Pericarditis: A Systematic Review. JAMA 2015;314:1498-506. [Crossref] [PubMed]
Cite this article as: Mackman S, McLeish T, Bhatnagar A, Jacobson N, Aranda J. Pericarditis in the immunocompromised patient: a case report. J Emerg Crit Care Med 2024;8:6.


